SARS-CoV-2 hijacks the Kynurenine Pathway
AhR Activation and Kynurenine Signaling: Implications for SARS-CoV-2 Pathogenesis
At some points of this particular substack, I will use crude language. In fact, probably in the next paragraph. The F word.
Given how people responded to merely tweeting about the article (without the extensive breakdown and everything else below) I decided before publishing to add how you may deal with what we will discuss.
While you can modulate the receptor we are about to discuss, it is too important to “mess with”, and there is extensive evidence of the positive effects of modulating the Kynurenine Pathway itself.
Fasting, being in Ketosis.
NAC+Glycine
Pyridoxine (Vitamin B6) (Thanks Paul)
Thiamine (although very indirectly)
Melatonin (directly, such as B6 and GlyNAC)
Metformin (possibly Berberine)
But above all else the bet Tryptophan/KP fix is EXERCISE.
Basically, the supplements and steps I suggest for dealing with SARS-CoV-2 and many other conditions.
There are many other avenues of inquiry and research in regard to SARS-CoV-2, but lately, scientific publications took an interesting turn in regards to my perspective and observations on SARS-CoV-2 and its complex interactions with the human body, evidence and understanding of how things work is necessary for both scientific advancement, and to propose a viable hypothesis, and at a “personal level” to keep a track record. In this regard…
Finally, after two long fucking years I have one of the answers I have been looking for, extensively. It has been no secret the amount of focus and work I put into an aspect not widely discussed, a significant pathway that SARS-COV-2 hijacks to manipulate the host’s metabolism and immune system, the Kynurenine Pathway. Besides various mentions and links to papers in “random” substacks I have quite a few that are solely focused on that. Some of the “random” ones are more pertinent to the future than others.
At first, there was an overrepresentation of the KP in more than one subset of Long Covid patients, followed by evidence of its activity and effects on both severe and critical patients, soon followed by evidence that this particular pathway is active at every degree of infection, but many factors influence the “end result”. It bears a disproportional weight in a lot of everything I wrote, and it is the de facto perfect example of a biological double-edged sword (or as I often refer lately, very contextual effects).
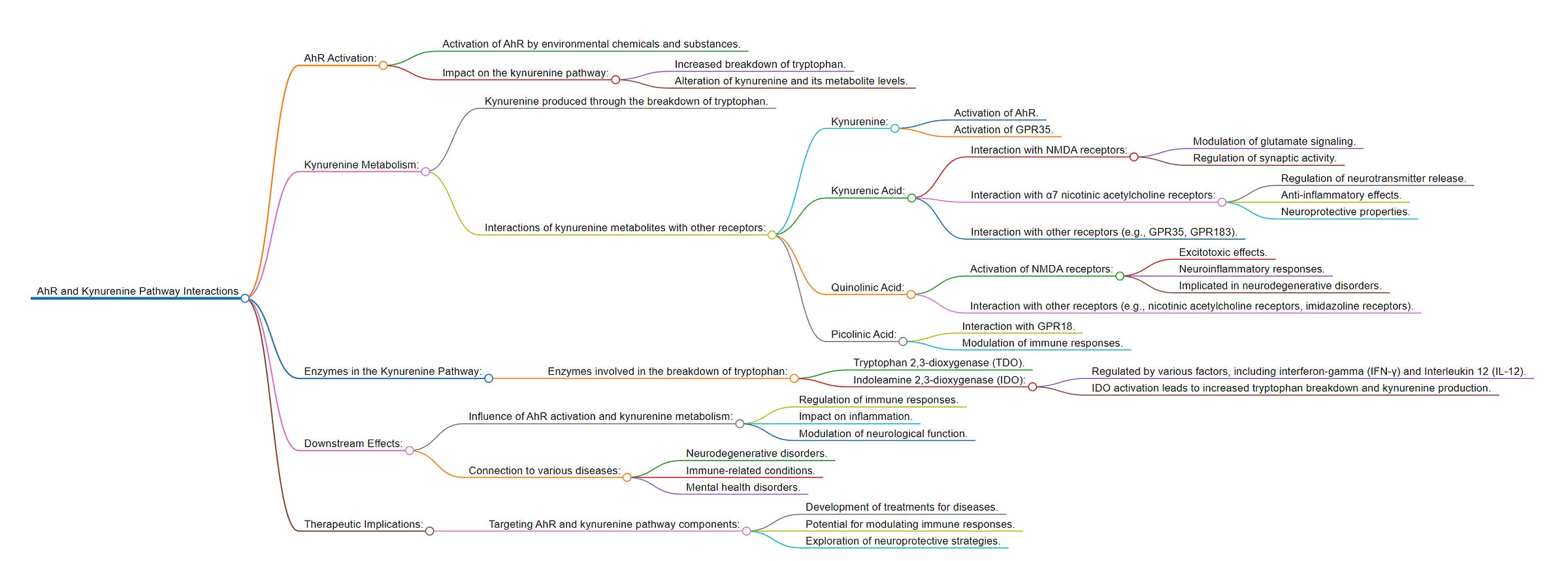
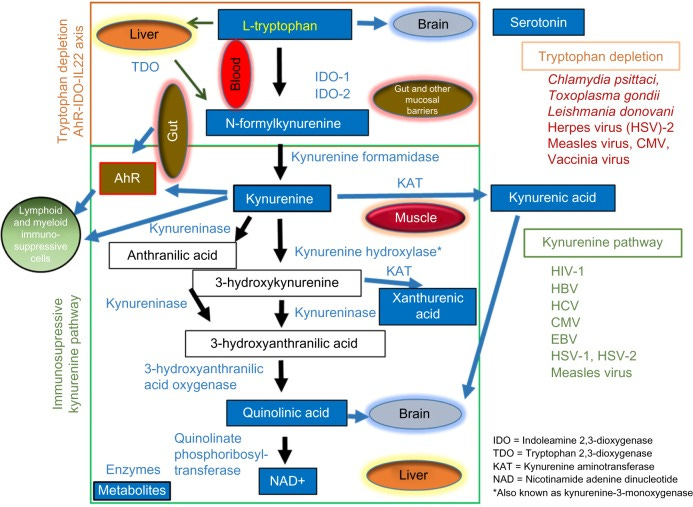
Here is a graph I generated for my readers to better understand the content in the substacks above, and as a “refresher”. Also helpful for visual learners (thanks bro who helped me).
The substack below talks about the AhR in the context of the mRNA platform (especially vaccines), and the evidence for its highly inflammatory profile.
Aryl hydrocarbon receptor is a proviral host factor and a candidate pan-SARS-CoV-2 therapeutic target
The aryl hydrocarbon receptor (AhR) is a ligand-activated transcription factor and a nuclear receptor. Similar to other nuclear receptors, the activation of AhR is ligand-driven. In the absence of ligands, AhR is localized in the cytoplasm as a part of the molecular chaperone complex. fter activated by the ligand, it enters the nucleus and forms heterodimers with AhR nuclear translocator and specifically binds to AhR reaction elements. This interaction between AhR and its ligand activates the expression of a series of genes downstream of AhR, such as cytochrome P4501A1 (CYP1A1) (13). The AhR is widely expressed in innate and adaptive immune cells. AhR has a variety of physiological functions, including the regulation of immune and inflammatory processes and environmental responses.
The activation of indoleamine 2,3-dioxygenase 1 (IDO1) causes immune cells to release kynurenine (Kyn), a tryptophan (Trp) metabolite, and an endogenous ligand that activates AhR, which interferes with protective immunity (14–17).
This entire paragraph is significant, not solely on how it explains how AhR works (interpret ligand as a signal for a cell function, in this case AhR needs something to activate it), but where it is located. The location of the receptor and the paragraph bears important information and dynamics to understanding how the virus impacts the immune system long-term. And the second paragraph is the most significant dynamic here.
SARS-CoV-2 infection activates AhR signaling in an IFN-IDO-Kyn–dependent pathway
To understand how SARS-CoV-2 induces AhR signaling, they performed RNA sequencing from macaques lung tissue infected with SARS2 using different types of lung cells for this test, and the results of the sequencing showed increased expression of upstream gene IDO1, and downstream transcriptional targets CYP1A1 and CYP1B1 of AhR (both of these genes are responsible for many cell function but in this context, the main ones would be inflammation, oxidative stress and at a lower level DNA damage), which they further tested using other models. While other types of pathways and genes are obviously expressed during infection, they decided to choose AhR signaling in the context of promoting viral replication.
The next step is studying the molecular pathway of AhR signaling, since IFN (Interferons) can activate AhR signaling, the ey detected both IFN and IFN-stimulated genes (ISG) in cell lines, finding that specific genes were significantly upregulated, with the more significant being IFNB1 (gene responsible for IFN Beta… interestingly enough this protein plays a significant role in Multisystem Inflammatory Syndrome in Children).
Next, the researchers explored the molecular pathway of AhR signaling activated by SARS-CoV-2 infection. They investigated the expression of interferon (IFN) and IFN-stimulated genes, which are involved in antiviral responses. The results showed that these genes were significantly upregulated in SARS-CoV-2-infected cells. Additionally, when the cells were treated with IFN-β, they observed increased expression of AhR pathway genes, suggesting that IFN-β activates AhR signaling. In the words of the authors themselves.
These results indicated that SARS-CoV-2 infection induced IFN production, which further promotes Trp metabolism to produce Kyn. As an endogenous ligand of AhR, Kyn promotes the entry of AhR from the cytoplasm into the nucleus and initiates the expression of AhR transcription target genes.
Together, these results demonstrated that SARS-CoV-2 infection activates AhR in an IFN-IDO-Kyn–dependent pathway.
Following by knocking out (removing something from cells) AhR, and testing the levels of N protein and viral titers they found that in the cells without AhR, N protein and viral titers were significantly lower than “wild-type” meaning cells containing the receptor. Demonstrating that AhR plays a crucial role in infection, and increasing AhR activation increases N protein and viral titer.
AhR boosts SARS-CoV-2 replication by limiting IFN-I response and up-regulating ACE2 transcription
Being a nuclear receptor and a transcription factor (regulating genes) AhR also plays a role in modulating, in this case inhibiting the expression of Interferon I (IFN-I). IFN-I plays a crucial role in inhibiting viral replication, including SARS-CoV-2, given their findings they decided to explore the potential role of this relationship between the virus, the receptor, and interferon-I. When AhR was inhibited in the cell lines, the expression of IFN-I and ISGs increased in the cells infected with SARS-COV-2, furthermore inhibiting AhR led to a decrease in the level of SARS-CoV-2 viral RNA. These findings strongly suggested that AhR promotes viral replication by limiting the host's antiviral response mediated by IFN-I. In essence, AhR activation acts as a barrier to the effective antiviral mechanisms triggered by IFN-I.
Remarkably enough, they also observed that viral replication was restricted by IFN-I-Independent mechanisms, to this observation they tested the virus infection on cells (Vero in this case) deficient in IFN-I, which was confirmed by quantifying virus production. Inhibiting IFN-I signaling with another type of inhibitor (Jak1 inhibitor) also attenuated viral replication. Thus, AhR appears to interfere with IFN-I-independent antiviral mechanisms as well.
As their last step, and one with the most impact since the start of the pandemic, they sought to find if there was a relationship between AhR and ACE2, finding that AhR directly regulates the transcription of the ACE2 by binding to its promoter, therefore demonstrating that AhR activation causes the upregulation of ACE2, which potentially may worse the pathology.
A simplistic summary, AhR enhances SARS-CoV-2 infection by limiting IFN-I-dependent antiviral mechanisms and up-regulating the expression of the ACE2 receptor.

You should refer to the graph I created as many times as needed, if you have an interest in understanding the complex relationship between KP and everything discussed you should save it. The findings of this paper are very significant because they demonstrate mechanistically the virus hijacks the KP pathway using the IDO pathway.
Outside the scope of their proposition, this is significant. I will make it short and simplified, Kynurenine and diabetes, insulin resistance, cancer, the virus itself (the stalk of the spike) directly affect the expression of α7 nicotinic acetylcholine receptors on the cell surface, but Kynurenic Acid (KYNA) does the same, with many implications for both neurological and nervous system diseases (hallmarks of Long Covid, and damage from the infection). Among its many other roles, it also plays a very significant role in chronic infections.
Before my closing thoughts, a very recent paper on Long Covid was published in Nature.
Here we evaluate the serum proteome in samples, longitudinally collected from 55 PASC individuals with symptoms lasting ≥60 days after onset of acute infection, in comparison to samples from symptomatically recovered SARS-CoV-2 infected and uninfected individuals. Our analysis indicates heterogeneity in PASC and identified subsets with distinct signatures of persistent inflammation. Type II interferon signaling and canonical NF-κB signaling (particularly associated with TNF), appear to be the most differentially enriched signaling pathways, distinguishing a group of patients characterized also by a persistent neutrophil activation signature. These findings help to clarify biological diversity within PASC, identify participants with molecular evidence of persistent inflammation, and highlight dominant pathways that may have diagnostic or therapeutic relevance, including a protein panel that we propose as having diagnostic utility for differentiating inflammatory and non-inflammatory PASC.
In a subset of Long Covid patients with persistent symptoms, the researcher found peculiar persistent responses, some normalize within 6 months post-infection, but in certain patients, failure in Type I IFN (the one we just discussed above) leads to momentary activation of Type II Interferon, but in Long Covid patients examined here this Type II responses as sustained for weeks, and they argue one of the reasons could be the persistence of viral fragment in the body.
The reason I bring this paper up, besides its importance on its own is two-fold.
IL-12/IFN-gamma axis is a known modulator of the IDO pathway, leading to both immune suppression and inflammation, and abnormal activation of the Kynurenine Pathway, shifting Tryptophan to the “wrong side”. It is, effectively, a fairly well-known metabolic trap
The genes that are regulated and shifted with the persistent or high levels of this axis are one of my main arguments against the mRNA vaccine and are responsible for a lot of diseases of many etiologies
Here we are presented with a Hit and Run effect with a “real” mechanism behind it. Using different pathways, the mRNA vaccine will induce similar effects as the viral infection (AhR, IL-12/IFN-gamma, things discussed here), therefore sensitizing the body towards a “secondary hit”, the viral infection itself, compounding the effects of the first hit.
Tryptophan is a very important amino acid, one could argue the most important one for life, I plan to briefly cover why and how soon, but the findings discussed here demonstrate how and why tryptophan is being deprived in many, explaining many symptoms, from mild brain fog and insomnia for a few days to a percentage of Long Covid patients and autoimmune/thyroid issues. This is already long enough.
For the first time, we are finally reaching a point where a formal hypothesis could be proposed, highly complex, but now with enough evidence to back it up. (I won’t do it FYI)
If you choose to support this work in whatever form, thank you !







Been waiting for this. Many thanks, so damned complex.
It's one huge clinical trial, isn't it?
Got here late- fantastic!