A paper I didn’t think I would ever write about was published today, but before I went to write about it, I wanted the “receipts”. This is as far as I found it, my private (encrypted) chats with the person I collaborated with in the PAID hypothesis, therefore basically the entire backbone of all my Substack are now lost.
I would also like to leave a formal big thank you to that person for always pushing me to learn more and more. No easy answers.

If you want a deeper understanding of the mechanism of what we are about to discuss, here is a brain, one focusing on the metabolic aspects, with this one being fairly important from a mechanistic perspective. The one below touches on another mechanistic aspect of this, and how it fits in the overall hypothesis.
I do admire the honesty of the authors in which, unlike many other papers, they don’t state they found a new wheel, but rather that mechanism for neurodegeneration in regards to SARS-CoV-2 remains unknown. Yet they made strides to partially solve this.
Here using humanized transgenic mice expressing ACE2 authors decided to test the long-term implications of SARS-CoV-2, and directly injected a low quantity of the virus directly into the brain (this is one of the reasons I will never ever get any type of vaccine intranasally, it goes directly into the brain now, because BBBs are disrupted by many factors).
The list of genes in the supplementary data is quite extensive, but in the paper, they focused on specific genes, and here a few of the most important ones were ones linked to antiviral immune response and of interest aging (as in…inflammaging), all the finding suggesting persistent neuroinflammation, but I would go further and they are suggesting more immune and metabolic dysfunction in the brain, and I will make my case further down.
From the upregulated genes, Trem2 is somewhat of a compensatory one, attempting to fight back some of the inflammation in certain neurodegenerative conditions, while Gfap is mostly a pro-inflammatory one, leading to loss of function, plasticity, and higher levels of oxidation. Downregulated genes are, as one would be expected, linked to neurodegeneration, they feed the dysfunctional state that leads to “bad” proteins aggregating in the brain. Unexpected to the authors and myself, Amyloid Precursor Protein (APP) was unchanged… this will be important shortly. The closing remark in green is the most important one here.
To further understand the potential mechanisms for what we just read, they tested if SARS-CoV-2 Membrane Protein had any role in this. Before going further we need to make something clear because a lot of readers won’t be aware.
γ (Gamma) secretase is a complex (as in many parts) protease (something that helps break down other proteins) made up of 4 different proteins, among some other regulatory functions. Among the “proteins” it does this, the most well-known is APP.
The γ-secretase complex, comprising PEN-2, APH-1, PS1 and NCT, is a critical membrane complex contributing to Aβ production in Alzheimer’s disease (AD) pathogenesis8.
They found that the Spike S2, but not S1 interacted individually with each of the proteins above, and not only that but also bound to all of them. By further testing, they found that the Spike S2, not the Membrane interacted with Gamma secretase, and further tests led to the finding that S2 directly binds to PS1 and NCT.
What does this mean in non-science speak ? If something binds (legos itself in the correct lego part of the other protein/cell) means it can modulate, therefore change activity, in this case, the S2 of the Spike Protein can directly modulate Gamma Secretase
Since they were using “old” Spike they decided to test Omicron BA.1 Spike, because as you can see, it had 6 mutations in the S2 section, and well…. the Omicron Spike S2 has a similar capacity to do the same as “old” Spike.
The next step was to test Gamma Secretase could cleavage (cut) Amyloid Precursor Protein, in which in this case there is only one site (location) in which Gamma can do it called APP C-Terminal 99 Fragment (APP-C99), and their results demonstrated that is, there was an increase production of AICD (APP Intracellular Domain) a byproduct of Gamma Secretase “cutting” APP.
By using transgenic mice (this one) their tests showed them that there was a high increase in soluble Aβ40 and Aβ42 (arguably the culprits of Alzheimer’s) after S2 delivery into the mice brain, showing that the S2 overexpression exacerbated the burden of Aβ, in simple terms, it would accelerate or worsen what was already there, also consequentially “flip” the necessary switches to develop neurodegeneration. Finishing their paper with this.
The Spike protein could function as an immune switch to increase γ-secretase activity and Aβ production and contribute to neurological changes in COVID-19 patients.
The summary so far, the expression and presence of S2 in the brain are enough to increase Gamma-secretase and Aβ progressing and enhancing the neurological losses present in a portion of people infected with SARS-CoV-2, and once again I must add, this will mostly be linked to severity and/or viral load, and the capacity of someone to effectively clear the virus in a timely manner.
Being clearly a problem because one of the hallmarks of SARS-CoV-2 is the production of APP after infection, related to the severity of the infection, and also your own metabolism, but the other side is the IAPP, known as Amylin, also severely overproduced in SARS-CoV-2 and it is more related to your metabolism, the liver, and your glycemic control.
With both of these present, IAPP and AB42, they can work in conjunction to further progress neurodegeneration, neural cell death, in simpler words, disease acceleration. This one is long enough but I wouldn’t be me if I half-assed this one, especially this one.
Gamma Secretase is a very important enzyme, very much.
Gamma Secretase is directly linked to the signaling and expression of IL-17 and Th17 immune cells differentiation by Notch, therefore the overexpression of this enzyme, allied with everything else we know about Spike and mRNA clearly points a highway towards Rome. Also worth noting blocking Gamma, suppressed the production of IL-12 and if you read the PAID hypothesis Part III you will know how significant that is and promoted the production of IL-10 something a good portion of Long Covid has a deficiency or is dysfunctional.
Mitochondria also have Gamma Secretase, and it participates in the process of mitochondrial metabolism and in the presence of APP and its byproducts and may contribute to mitochondrial dysfunction in neurodegenerative conditions. As I wrote quite a few times recently, all roads lead to Rome, and Rome has a bioenergy problem.
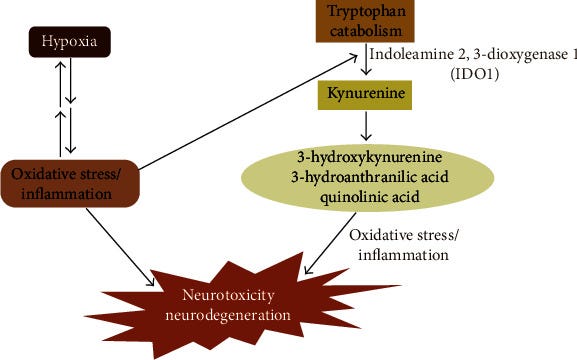
If you read some of the older pieces I linked you will find a common thread besides mitochondrial dysfunction inside them. HIF-1α. Hypoxia Inducible Factor-1 Alpha
Why is that important ? (I plan to cover this entire paper in the Kynurenine part II, but I will leave it here in case anyone wants to read it)
Hypoxia drives the metabolism of APP, leading to the amyloidogenic pathway, with Aβ protein as the end product [69]. Mattson [70] reported that this pathway could be a defense mechanism by increasing soluble neuroprotective APPα production. However, hypoxia favors APP metabolism through the amyloidogenic pathway, causing an increase in Aβ levels and not APPα levels
The positive feedback loop increases BACE1 levels and HIF-1α-induced genes, increasing amyloid-β protein production. This effect may occur because of the stabilization of HIF-1α, which Aβ also upregulates. Moreover, the fact that HIF-1α-deficient mice reduce BACE1 expression shows that HIF-1α is an essential mediator in BACE1 induction in hypoxic condition
Furthermore, energy or oxygen supply imbalance may activate various signaling mechanisms, including glutamatergic synapse formation, MAPK/PI3K-Akt signaling, and phosphatidylserine translocation, which play vital roles in oxidative stress and NDs. Indeed, HIF-1α plays a twofold role through gene activation, in the sense that this factor has to “choose” whether to protect or to kill the affected cells [109]. The regulation of HIF-1α, the exploration and internal control of the kynurenine pathway via the blockage or the expression of some critical metabolites, is a beneficial and potential medical target in neuroinflammatory and ND treatment. Taken together, the activation or inhibition of hypoxic intermediates with or without the kynurenine pathway metabolites could serve as novel therapeutic strategies for neurodegenerative disorders
HIF1α is a double edge sword in the presence of high expression of kynurenine in viral infections because it will attempt to limit the production of kynurenine and the expression of IDO, which will modulate what happens to APP, in which case it trades neurotoxicity for plaque accumulation and neural death, and this is another one of those complex loops this thing creates.
I will openly say there is more hidden in the S2 portion of the virus than we are aware of. In fact, this was brought to my attention by the friend I mentioned at the start of this piece.
The Spike Protein S2 binds to Estrogen Receptor Alpha. Estrogen directly affects Kynurenine Pathway.
And finishing this with a tweet from almost 1 year ago.
Biggest appreciation for all supporters here and in Kofi you make this work possible, and the ones who share my posts !










I came to this late. Trying to catch up and digest. Your late statement is breathtaking. With every bit I understand; the more concerned I become. Weaponization of homeostasis, acceleration of disease. We see that everyday whether anecdotally or all cause mortality. Thank you for keeping on, keeping on.
Great work helping to solve the mystery of what can happen to the cleaved S2. Nothing good. And funny you mention HIF:
...Ubiquitination is described, as is evidence for the HIF inhibitory effects of both lactoferrin and ivermectin. This is important for working synergistically with doxycycline to help avoid resistance from cancer cells.
https://doorlesscarp953.substack.com/p/repurposing-of-the-antibiotic-doxycycline