Serotonin "deficiency" by multiple mechanisms
SARS-CoV-2
I thought of leaving a caveat at the end of this article, for better coherence, but I will leave it here first, and it will only make sense after you read the entire thing.
I highly advise you, if your problem is Long Covid or a similar situation, to DO NOT supplement only one thing, in this case just Tryptophan or 5HTP. Researchers doing these awesome studies must stay focused and can cover every possible road, but most post-viral sequalae never come “alone”.
So you must start by addressing more than one causative agent. If you have developed fungal problems, you need one of the two (tryp or 5htp).
This one will be a little bit different than the last one, given how extensively I covered the subjects discussed, I will write in a more simplified manner here is paradoxically very complex. Proteins and how they interact are already complex, but when you add a systemic hormone that is a byproduct of a very chaotic metabolic pathway…
well, as they say, “Yeehaw !!!”.
A short summary of the article above.
So why Long Covid patients crash ? While there is no definitive answer so far, causative mechanism as the experts would say, some of the underlying mechanisms are evident if you do a “meta-analysis”, reading dozens of papers, sometimes in an interdisciplinary manner.
LC patients have less efficient and overall fewer mitochondria, more oxidative stress (I like to call it cell rust for easier understanding), and a lot more inflammation at baseline meaning by doing nothing at all they already have more inflammation than a normal person, change in their muscles themselves, their bodies can’t transport the energy well too, and their muscles have more amyloid deposits than a normal person from the get-go. Above all else, and significant here, their bodies don’t transport oxygen that well.
With these variables, LC patients experience sudden crashes after certain levels of exertion, something I have experienced myself multiple times before 2019. In the case of post-Covid symptoms, long-term sequelae, and all the paradoxical changes, there is often a common denominator, one that can be found at every twist and turn. Every likely hypothesis, mechanism, or pathway either finds itself around, in, or ending up there.
Tryptophan and its complex metabolism. The Kynurenine Pathway. And the end product. Serotonin. There are 9 amino acids that are not synthesized by our bodies (histidine, isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan, and valine), I would add cysteine too (early bacterial life couldn’t have existed without cysteine). But I refer to Tryptpohan and Cysteine as “primordial” amino acids. Life wouldn’t exist without them (life wouldn’t exist without these amino acids, but I argue evolution would be hindered, severely, without these two).
A simplified visualization of most of this article.
Before delving into both papers, I first need to a short explanation of why serotonin is important. If tryptophan and enzymes the body uses to break it down, and the byproducts of this breakdown (called metabolites) can directly affect numerous cells and pathways in your body, one can deduce the end-product would do the same. In a paradoxical and very simplistic way, Tryptophan, the enzymes, and Kynurenines can be anti-inflammatory, and serotonin on the other hand can end up being very inflammatory. The entire metabolic pathway is the perfect definition of a double-edged sword.
You can find the paper here.
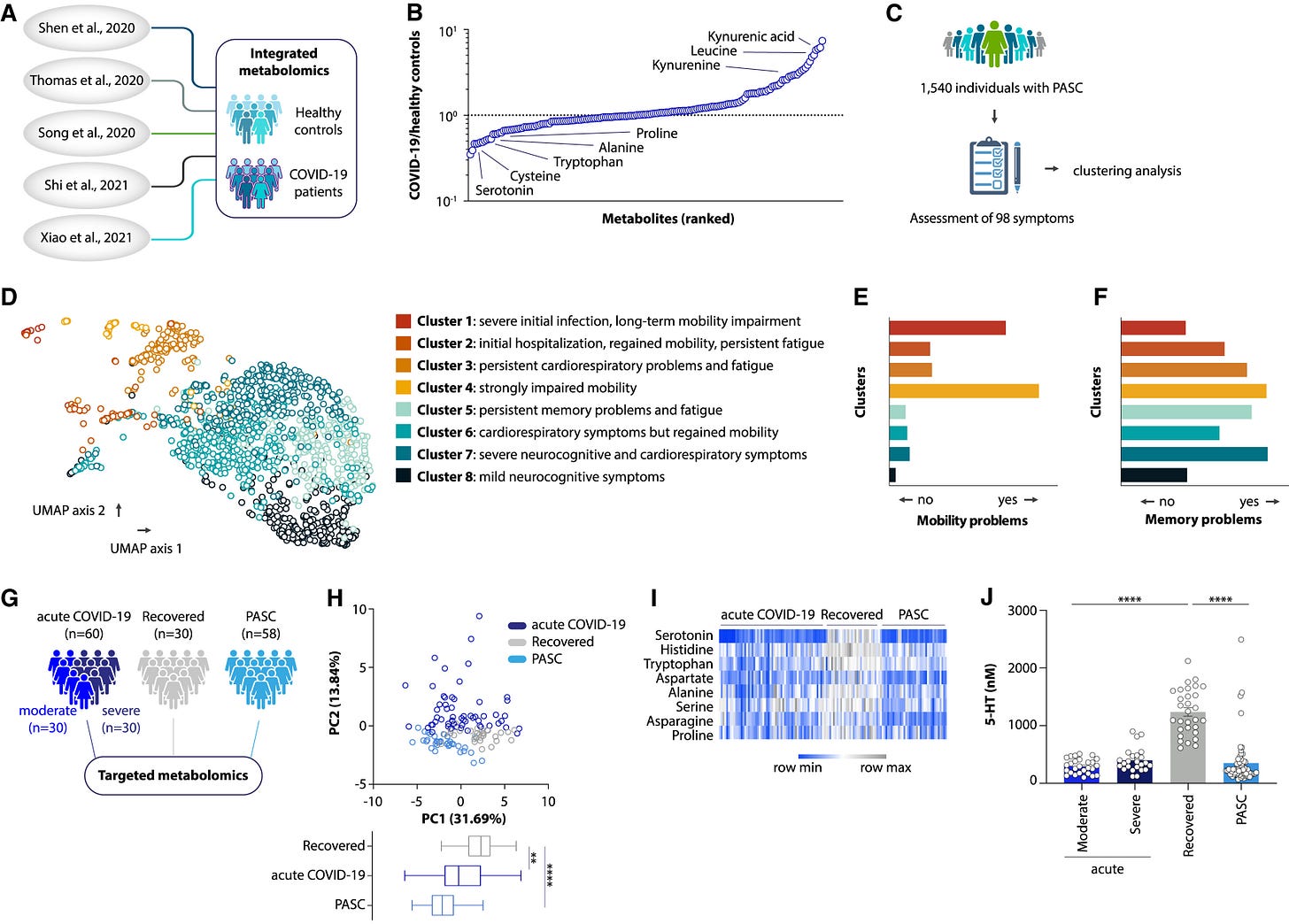
To start their exploration of the potential roles of serotonin itself in PASC/Long Covid (LC from here on) the authors decided to define a consensus metabolomics in LC, metabolomics is measuring large amounts of metabolites and measuring how much or how little of each there is. A comprehensive snapshot of our metabolism.
They integrated metabolomics datasets from previously published studies, and among the metabolites most strongly altered during acute infection were amino acids. By analyzing the symptoms of a large group (1540) LC, the authors defined 8 clusters of LC, these are what I call subsets/subtypes.
By doing metabolomics tests on 58 LC patients, comparing them to 60 acute Covid and 30 recovered without symptoms the authors observed that the metabolite profit in LC was distinct from people who recovered without symptoms. Among the metabolites depleted during and after acute infection, the most affected one was serotonin. In a post-acute infection, serotonin levels were predictive of whether someone fully recovers or develops long-term sequelae.
Serotonin is central to a large number of functions of the body, inside and outside the nervous system/brain, so the authors now wanted to understand if this serotonin depletion was unique to SARS-CoV-2 or other viral infections led to a similar decrease. As this was observed, other systemic viral infections also lead to serotonin depletion.
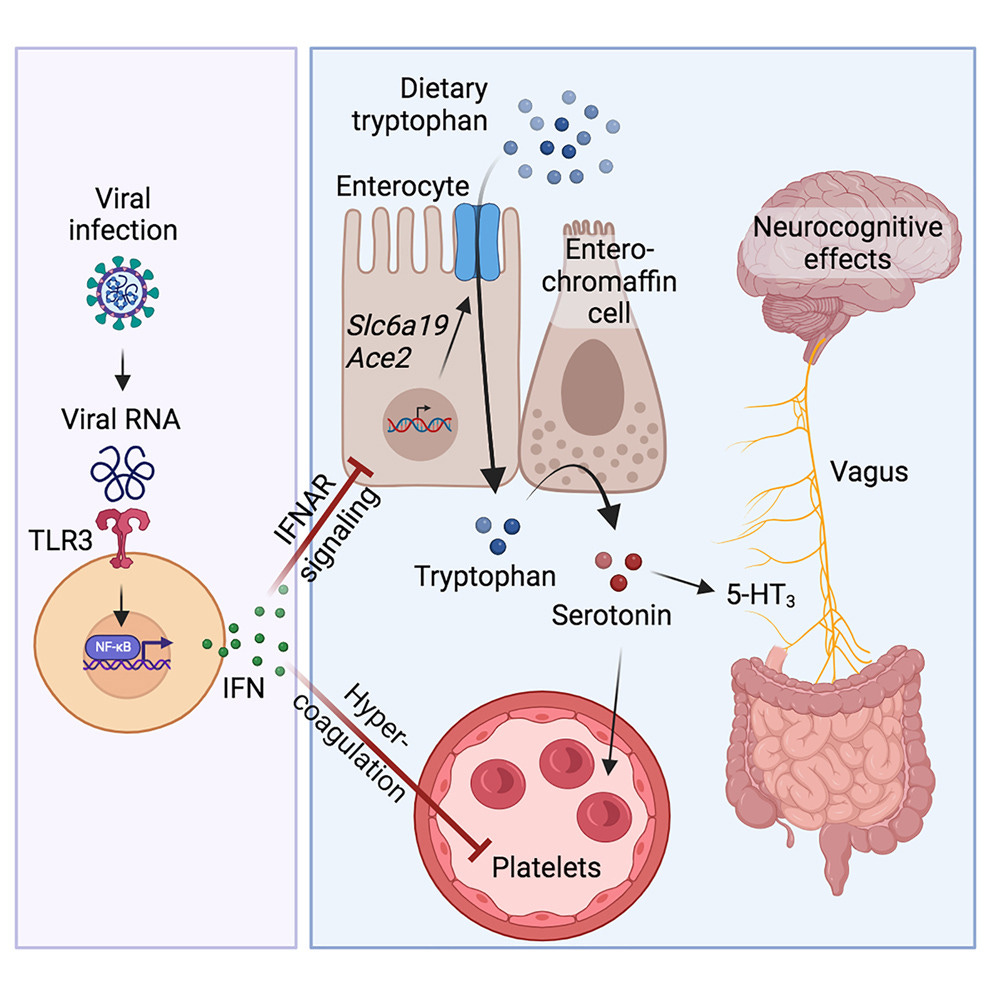
Viral inflammation decreases plasma serotonin levels
By infecting mice with human ACE2 receptors with different SARS-CoV-2 variants they observed a reduction in serotonin levels, and the same was observed with other viruses. Next, given the likelihood of viral persistence being one of the causes of some LC, they tested the hypothesis and found that chronic infection also reduces serotonin.
If the chronic infection can induce long-term decreased levels of serotonin, they tested if the viral inflammatory process itself, absent of virus replicating can be one of the causes, by using poly(I:C) a TLR3 agonist. Using poly(I:C) results in lower serotonin levels in both the plasma and in isolated platelets, platelets are the major reservoirs of serotonin around the body. 1 week after ceasing the use of poly, serotonin levels normalized, indicating that the process itself is reversible.
A common denominator in these 3 variables, viral infection, chronic infection, and engagement of TLR3 has Type I Inferon signaling as the result. By blocking Interferon signaling they prevented serotonin reduction, moreover by using genetically engineered mice without TLR3 or one of the main interferon-stimulated genes (STAT1) it was observed the mice were resistant to the process of serotonin reduction by the mechanisms above.
Viral inflammation blocks intestinal tryptophan uptake
The majority of serotonin, like other neurotransmitters, is produced in the gut, where tryptophan is absorbed. After analyzing different groups of patients, they observed that the cause of low serotonin is most likely caused by low tryptophan, this was also observed in their chronic infection and poly models, and feeding a low-tryptophan diet had the same effect on plasma serotonin as using poly(I:C).
The two main causes for tryptophan deficiency in normal “healthy” adults are usually reduced intestinal absorption or a lot of kynurenine pathway activity. Here is where the authors surprised me because in mice lacking IDO1 (the main kynurenine “thingie”) lower serotonin levels were still present, same with TDO2, another arm of the Kynurenine Pathway.
After a series of tests, the authors found this inflammatory process itself affects the genes responsible for amino acid transport, and this process directly affects ACE2 which is responsible for tryptophan absorption in the gut. Gut ACE2 is also responsible for regulating glucose metabolism. Interestingly enough blocking NF-κB (which is a downstream target of TLR3, a very potent factor) blunted the downregulation of ACE2. Nicotine as I wrote 2 years is a potent NF-κB inhibitor, and this is why certain Long Covid and other people may experience improvements in their symptoms.
It was also observed that the presence of viral components in the gut possesses similar detrimental effects on ACE2 levels, therefore directly affecting amino acid absorption and serotonin levels, in this case, some people who have either the virus in their gut or viral fragments will experience this feedback loop. I will leave the next part to the authors themselves.
If tryptophan uptake was abrogated by poly(I:C) treatment, tryptophan supplementation should elevate serotonin levels even during viral inflammation. To corroborate this, we used a diet containing a glycine-tryptophan dipeptide, which bypasses the need for B0AT1 and enables tryptophan uptake via dipeptide transporters. This diet compensated for impaired uptake in poly(I:C)-treated mice and led to an increase in both tryptophan and serotonin levels in systemic circulation . Similarly, supplementation with the serotonin precursor 5-hydroxytryptophan (5-HTP), which bypasses the requirement for tryptophan, rescued serotonin levels in poly(I:C)-injected mice. Collectively, these data demonstrate that viral-RNA-induced inflammation impairs intestinal tryptophan uptake, which causes systemic serotonin depletion.
Their last finding was in regards to how viral inflammation impairs serotonin storage, but this part is very “tricky”, in their model this occurs by the pathway described, but the proposed mechanism is correlated with platelets function, the same pathways that lead to lower serotonin, lower amino acid absorption also can induce platelet destruction. Tryptophan supplementation had no effect in restoring platelet counts, so the mechanisms being lower serotonin and this are distinct.
Upon platelet hyperactivation and too much clotting (hypercoagulability), the transport of serotonin throughout the body is severely compromised, this leads to “free” serotonin “floating” around your body which becomes the target of MAO (MonoAmine Oxidase, an enzyme responsible for “cleaning” neurotransmitters). It is one of the primary reasons I advised for the last 4 years to not induce MAO activity in regards to Covid.
Lastly, the authors tested the hypothesis that low serotonin impairs vagus nerve signaling and affects memory function.
Given the dual role of ACE2 as both a mediator of intestinal tryptophan absorption32 and a receptor for SARS-CoV-2,76 it is possible that virus-induced receptor internalization augments the effect of interferons on ACE2 downregulation and serotonin reduction. In principle, however, none of the mechanisms described in this study are unique to SARS-CoV-2 infection. Indeed, reduced serotonin levels have been reported in other settings of viral inflammation, such as dengue virus infection,77 which is the trigger of another post-viral syndrome.78 The connection between serotonin reduction and vagus nerve dysfunction may thus be relevant beyond Long COVID. The fact that low serotonin levels are also found in non-viral conditions characterized by elevated interferon levels, such as systemic lupus erythematosus or multiple sclerosis,79,80,81 suggests that the pathway described in this study may even apply beyond viral infections.
Given how extensive and perhaps somewhat complicated even at the current state is, I will briefly cover the next part, because it is another side of this 20-sided dice, and do what I said I would connect most of the last published articles to what is described here. This was another surprise I had in 2023, and a paper I lost multiple times in crashes… thankfully I guess.
Another visualization of the article below.
Prolonged indoleamine 2,3-dioxygenase-2 activity and associated cellular stress in post-acute sequelae of SARS-CoV-2 infection
Findings
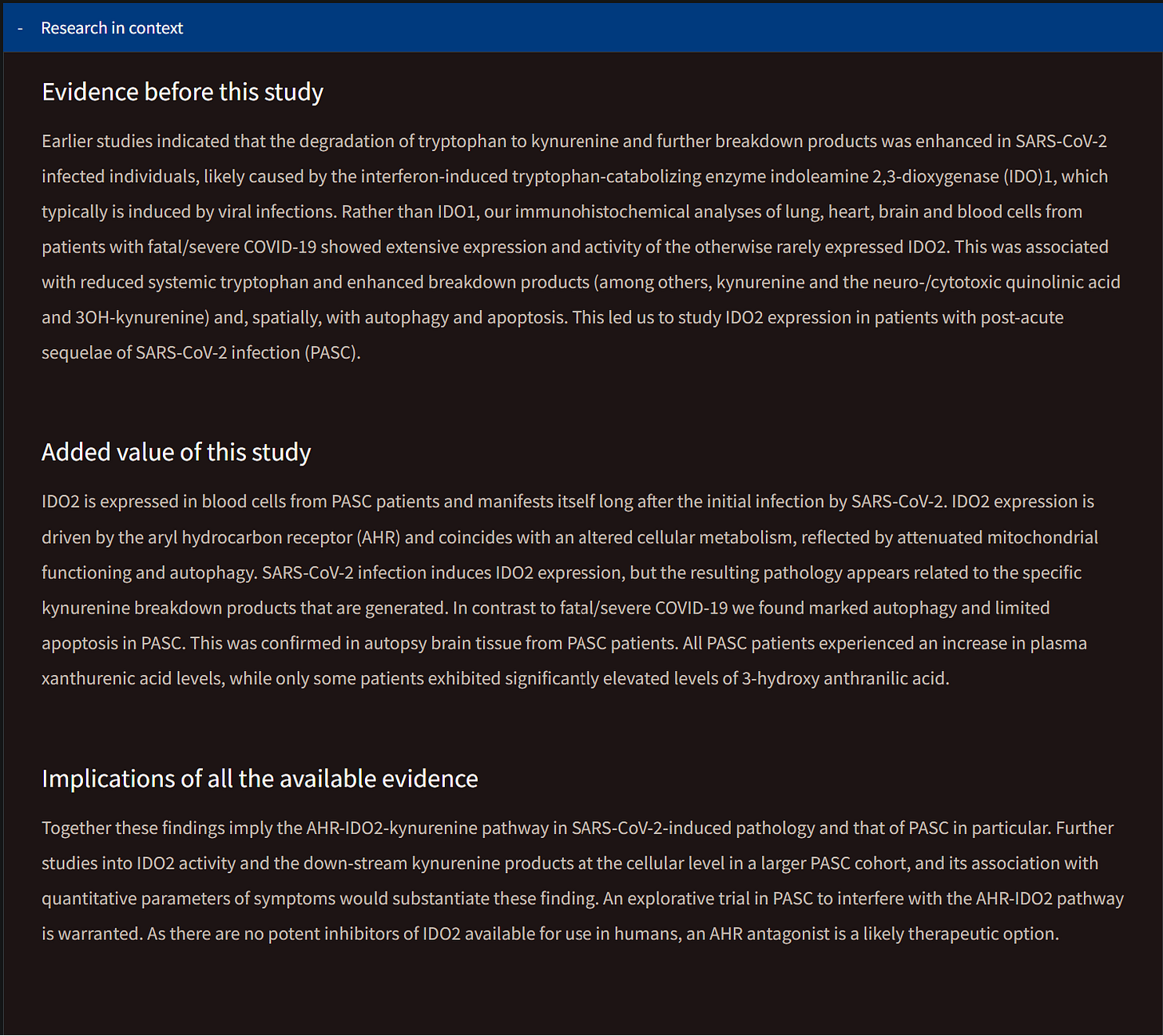
IDO2 is expressed and active in PBMC from PASC patients, as well as in brain tissue, long after SARS-CoV-2 infection. This is paralleled by autophagy, and in blood cells by reduced mitochondrial functioning, reduced intracellular levels of amino acids and Krebs cycle-related compounds. IDO2 expression and activity is triggered by SARS-CoV-2-infection, but the severity of SARS-CoV-2-induced pathology appears related to the generated specific kynurenine metabolites. Ex vivo, IDO2 expression and autophagy can be halted by an AHR antagonist.
Interpretation
SARS-CoV-2 infection triggers long-lasting IDO2 expression, which can be halted by an AHR antagonist. The specific kynurenine catabolites may relate to SARS-CoV-2-induced symptoms and pathology.
To my astounding surprise, this research group found something very unexpected, especially in LC patients, the long-term expression of IDO2. What you normally see referred to as IDO, is most likely IDO1, because it is the most abundant one whenever your body “needs” it. After all, it is the most efficient among them.
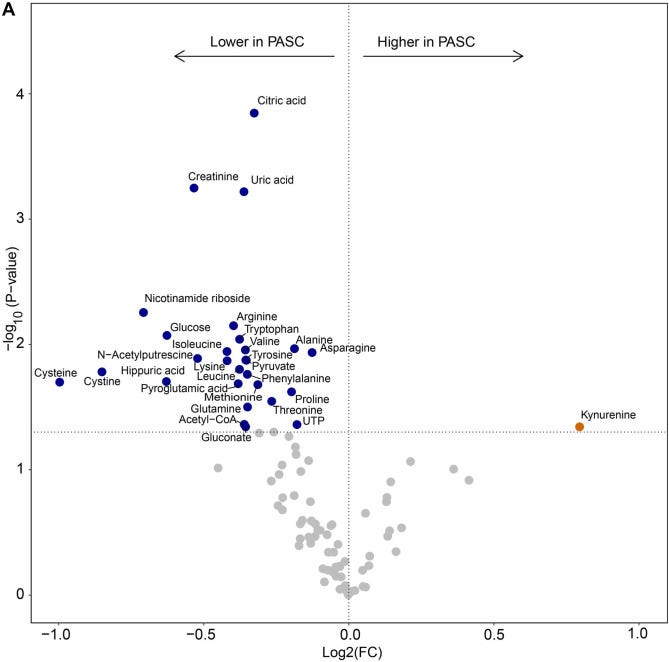
All groups of SARS-CoV-2-infected individuals had metabolites of the kynurenine pathway present systemically, markedly contrasting with reference values. However, there were marked differences between the groups for specific metabolites. In fatal/severe COVID-19, the researchers found the highest levels for kynurenine-metabolites (3OH-kynurenine, quinolinic acid, kynurenic acid), whereas levels of tryptophan and 3OH-anthranilic acid were lowest in comparison to the other groups.
In PASC patients, levels of xanthurenic acid were highest, and, although not significant, the median 3OH-anthranilic acid level was highest in PASC patients (up to 106-fold higher than the mean reference value; even though 5 out of 14 patients had no detectable 3OH-anthranilic acid) as opposed to 7.5-fold higher in fatal/severe COVID-19 (where all had detectable 3OH-anthranilic acid).
Even in patients who were either hospitalized with no or only mild remaining symptoms or were never hospitalized, increased median levels of 3OH-anthranilic acid were found. It is worth noting that patients in all groups had enhanced systemic kynurenine and downstream metabolites, even though the actual infection was at least more than two months prior to blood sampling, and even a median 294 days for PASC patients.
In a similar fashion to the substack referred to at the start of this article, on how and potentially why Long Covid patients experience a “crash”, the authors here found that the expression of IDO2 and the changes in other amino acids directly affected how much extra capacity and how much oxygen the mitochondria was using.
By analyzing more of the data the metabolites citric acid, glutamine, cysteine, arginine, and cystine, were the most important discriminators between high and low OCR (oxygen) and SRC (extra energy) values. Interestingly enough, while complementary the findings in this paper can also stand on the opposite side of the first paper we went through.
The Spike Protein can likely interact with the Aryl Hydrocarbon Receptor (AhR), and given this is a nuclear receptor (inside the cell, at the nucleus or cytoplasm) they are extremely significant and important and often interact with other nuclear receptors, these receptors regulate numerous functions. The kynurenine-AhR axis, even when added to any other protein is a self-perpetuating loop, this is one of the main reasons it is so important.
The long-term expression of IDO2 is of concern not only because it shifts the body’s immune response, but as the Kynurenine Pathway contributes to cancer, so can IDO2 aid in cancer progression. In many types of cancer IDO2 will aid in cancer progression. IDO2 can also exacerbate (accelerate) autoimmune disorders, in a similar fashion to the KP.
The dual-edge and closing remarks
Kynurenine and its metabolites can act as both pro and antioxidants, imagine as the backup plan in case something else goes wrong with the primary system (Glutathione). Kynurenine itself can act as an antioxidant on its own, in an attempt to increase Glutathione levels.
The metabolites of this pathway also contribute to the dysfunction of insulin and glucose metabolism, contributing further to a dysfunctional mitochondria state. Another feedback loop, since disruption in energy efficiency will promote the poor function of mitochondria.
All of the things discussed here affect your whole physiology, and especially your sleep pattern and quality. Sleep disorder and dysfunction will inevitably lead to Glymphatic dysfunction, the system your body uses to remove “bad proteins” from your brain. As supplementing tryptophan helps with what was discussed here, so does improve sleep quality.
Low serotonin, whatever the primary cause will also affect the glymphatic system, directly increasing the Amyloid Beta load in the body. This mechanism may help explain, at least partially, why patients who experience a “crash” after physical exertion have more amyloid deposition in their muscles both at rest and after exercise.
From this article I wrote months ago titled “The Importance of Proper Tryptophan Metabolism”.
Whey protein, one of the most researched supplements in scientific history, behind creatine alone, can help most people achieve proper tryptophan metabolism in the brain. It can also ameliorate anxiety and depression-like behavior by having an anti-inflammatory effect. Whey enriched with tryptophan enhances memory in MS. As a “joke” but with sound science behind it, whey has anti-viral properties.
This can explain why some people with Long Covid experience improvement in their symptoms because whey has a nice amount of tryptophan and other amino acids, and its absorption is made much easier than from diet, some whey brands will also contain the other amino acids disrupted by these mechanisms. It also explain why certain Long Covid patients develop fungal/yeast problems, such as serotonin-altering candida virulence
And here is one of the most important reasons any chronically ill person (or even healthy person) should supplement Thiamine (Vitamin B1).
While there are many distinct reasons the Kynurenine Pathway may be active even long-term in people suffering from chronic or even acute conditions, under certain circumstances, they may as well be a byproduct of the body attempting to compensate for the failure of its primary defense mechanisms.
Everything discussed here is of high importance, especially when I go back to continue discussing SARS-CoV-2 persistence and the complement system. But first I need to write about the Glymphatic System Part 2 and the article that will be very complex and most people won’t really like it, but it is of importance =P.
You can buy me a ☕ as a one-time thing. Thank everyone for your continued support !








Unrelated question. I decided to use the rest of my BPC-157. Good news, the pills don't "expire" even after the by date, but that is not reason.
Does anybody that uses or used BPC-157 experience becoming slower cognitively speaking. In blunt terms, you become dumber ? Or it is just me ?
It takes me a couple or three times to read these articles to understand it all. Question for you:
“MonoAmine Oxidase, an enzyme responsible for “cleaning” neurotransmitters). It is one of the primary reasons I advised for the last 4 years to not induce MAO activity in regards to Covid.”
Can you explain how “Not to induce MAO activity?”
Thanks in advance!