This article will be in similar style as recent styles, part science, part essay-ish.
First, we need to go back in time, to November 2019. Rumors in the image boards are vast and wide, and users able to navigate the Chinese web all whispers and rumors of a “new pneumonia that is wrecking certain Chinese hospitals”, by the first week of December, users all converged in a similar conclusion. A repeat of the Spanish Flu (an ancient meme embedded into our culture now) or a novel virus, which would most likely be a coronavirus given historical trends.
A lot of research was done in the boards between late November and March to April of 2020, so much research that it took the rest of the world, and researchers 18 months to catch up. If there is something the autists in imageboards always excelled, without parallel is pattern matching through vast amounts of data. A crowdsourced rudimentary biological machine learning if you will.
SARS-CoV had a lot of oddities, not only it suffered from lab escapes, but its outbreak wasn’t big enough to register in the epidemiologist’s radars, merely blips. But if you scoured the clinician’s notes and papers through the years you would find some unconnected points here, and there that raised more than one eyebrow. The Wuhan Strain was really aggressive, and from a forecasting perspective, I worked from the principle it would affect other pathogens, similar to how SARS-CoV did (and a handful paid attention until recently).
As years went by, independent and academic researchers alike were able to piece a lot of things together.
Among the most notorious effects of SARS-CoV-2 it is the ability to directly interact with other pathogen’s immune response. It can “wipe” out an important immunity axis, basically making certain people very prone to fungal infections. It can also directly disrupt biofilms, the little houses pathogens build inside you to hide from the immunity system. It forms different “clumps” of proteins with distinct effects. The last 2 cited articles are among the most important for our subject matter.
SARS-CoV-2 not only interacts but evolves and “picks up” advantageous mutations in the gut, in the microbiome. The presence of certain proteins from Staphylococcus Aureus accelerates the replication of SARS-CoV-2, thus tangentially helping its evolution. And this comes to the crux of the matter. If Staph Aureus is one of the biggest medical burdens, especially in acquired infections in hospital settings in the world, and has a significantly high mortality too, it causes sepsis and a whole host of long-term diseases. If it can help SARS-COV-2, did Covid help it in any way ?
There is a very particular, among the most important reasons for the long-term understanding of SARS-CoV-2, that I recently called it “a neo polymorphic chimeric toxin”, with a colossal emphasis on polymorphic, beyond current biology orthodoxy.
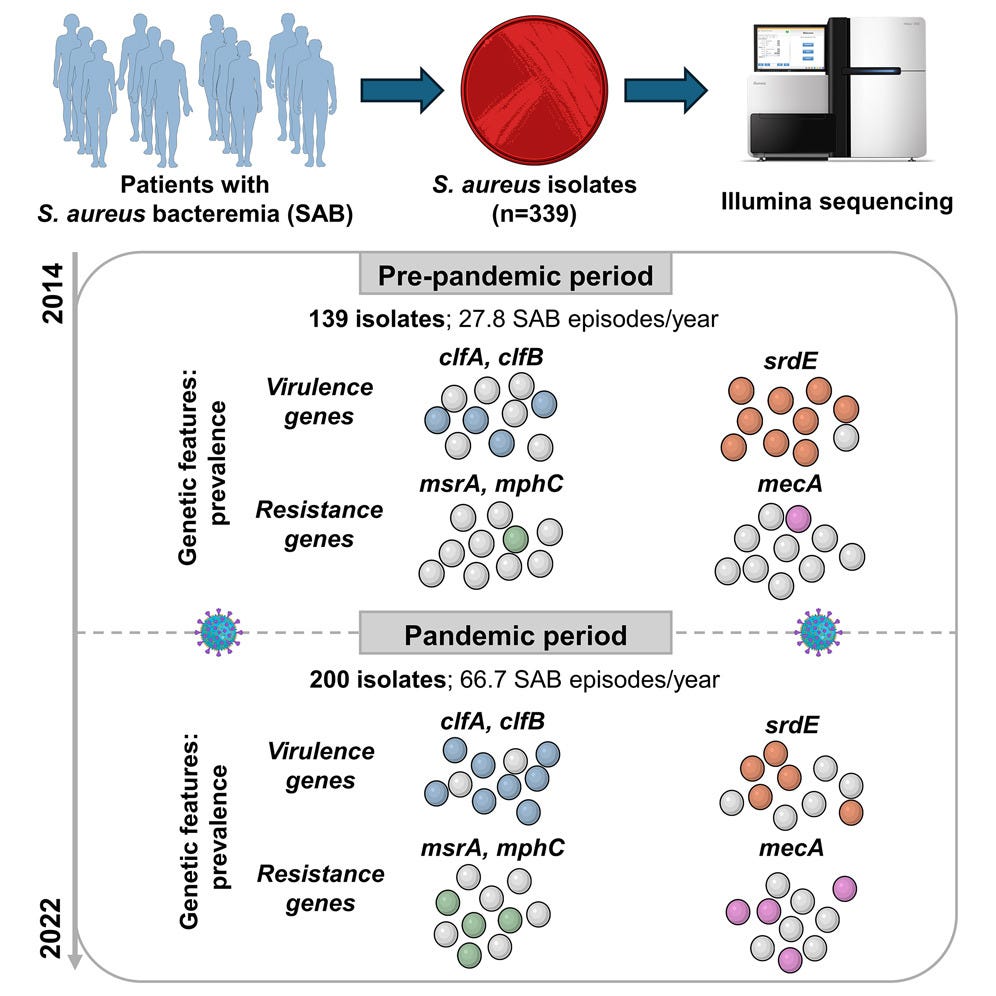
Genomic analysis of Staphylococcus aureus isolates from bacteremia reveals genetic features associated with the COVID-19 pandemic
By analyzing samples from an extended period (pre and post-pandemic), the authors were able to observe and track down differences in how each lineage behaved, the mutations they carried, and when since S. Aureus is well researched, what each of these mutations do, and what each different lineage contributed to in the case of patient disease and mortality.
Lineages like CC1 and CC8 had the highest mortality, CC15, 30, 46, 21 were the ones that created the longest (persistent) infections. CC15 and 45 were the ones associated with the most frequent “septic emboli”, clots containing bacteria floating around. Same with virulence and toxin-encoding genes, each lineage had a different set of these factors.
During the pandemic, there was a significant increase in S. Aureus bacteremia in their hospital, significant mortality in line with previous reports (around 20% to 30%), a significant increase of specific lineages such as ST398 which are resistant to treatments and often acquired in hospital.
Strains collected during the pandemic showed a increase in methicillin and macrolide resistance genes, including mecA, msrA, and mphC, which confer resistance to methicillin and macrolide antibiotics like erythromycin. Adjuvant treatments for Covid likely contributed to this, such as the widespread use of azithromycin.
The most interesting finding to me in regards to pandemic lineages was how specific clumping genes increased, clumping factors A and B (clfA and clfB), proteins involved in the pathogen’s ability to bind fibrinogen and both genes are known to directly interact with platelets and if that doesn’t ring any bells you must revisit my most recent articles. Covid increases fibrinogen production via multiple pathways, and S. Aureus adapted to it, and both genes had higher prevalence during the pandemic.
On the other side, they found a decrease in the prevalence of the sdrE gene, a gene that makes a protein that binds to Complement Factor H (CFH) a protein that is essential for the complement system. It helps S. Aureus prevent CFH from attaching to itself thus evading the immune system. It has been known since 2022 that the Spike Protein interacts with CFH (and also vWF). Redundancy in interaction means it becomes disadvantageous for the pathogen to carry the gene when both are present.
I could go on a large tangential tirade explaining how S. Aureus interacts with many of the pathways I have covered, and how it has cross relation with many effects of SARS-CoV-2, but in my mind, the most pertinent are superantigens, after all, SARS-CoV-2 literally has a piece of S. Aureus in the middle of its Spike Protein, the now infamous Staphylococcal enterotoxin B (SEB). A literal silent killer.
I like checking both sides of the aisle, anti and pro-mRNA to gauge where everyone stands, and how each side is concerned with certain topics. To my surprise months ago, I found a pro-mRNA molecular biologist PhD from Harvard stating something I brought up at the start of the text.
SARS-CoV-2 was affecting the evolution of all other pathogens, which is clear by now, not only S. Aureus, but many other pathogens have entirely shifted their epidemiological behavior, and there is a persistent increase in “rare” issues such as Mast Cells, Parvovirus B19 exploding after a few years of nothing. Extensive immune suppression and weeks on end of immune paralysis go unabated, undiagnosed.
And as a last point to make.


How does advanced HIV infection affect SARS-CoV-2 evolution ? The virus rapidly adapts and mutates to persist. Why does it matter ? Omicron was first found in Africa, the land of the immunocompromised. And Omicron quite literally replicated an “immune deficiency” state for a few months. S. Aureus can cause temporary T-Cell paralysis.
Evolution, such as greatness, at any cost. Such as my proposition of “SARS-CoV-2 as a neo polymorphic chimeric toxin”, I shall leave you with the following, clinical term. Adaptative immune impression, seen and proposed in regards to severe SARS-CoV-2 in 2021…
Thank you for your continued support ! You can buy me a coffee if you feel like it !






No hiking this weekend, so I will probably just work. Hopefully next week !
An update. Peptides are doing a lot of heavy lifting. It has been years since I don't experience significant pain on a daily basis (almost none lately). I am happy with my progress =).
I sort of want to crawl under a rock, but then there is probably some new chimeric pathogen lying in wait under the rock.