The Hidden Persistence of SARS-CoV-2
The Ambition to Become the Forever Virus
Your support of my work is greatly appreciated ! A small announcement in the comments too.
Among the many subjects related to SARS-CoV-2 that I hold the most interest in, one of the earliest was always “Latency/Persistence”. Very early on, as in March of 2020 given the research and what I observed at the time, I was already forecasting the virus would persist in a significant number of people, to the disagreement of most “experts” at the time.
Yet, if you spent any significant amount of time researching the first few SARS-CoV (the first) outbreaks, you would realize something was amiss, something smelled rotten in the kingdom of eerily odd SARS-related virus infections. Given how long I have written about this, here are two starting points that will inevitably lead you to other articles.
Simple put, in the last 18+ months there has been enough evidence that not only Spike Protein, but the virus and its materials persist, and we march on. While I spent most of the last few days researching the mechanism (the complicated stuff I usually write about) I will try to bridge the gap and merely present the information, an inevitable following article will cover a few of the potential mechanisms.
A visual summary of the entire article.
Prevalence of persistent SARS-CoV-2 in a large community surveillance study
The first study uses extensive data, almost 94.00 high-quality samples collected throughout the UK, samples were part of the Office for National Statistics COVID-19 Infection Survey (ONS-CIS), gathered between November 2020 and August 2022. How researchers track “persistent infections” is somewhat easy to understand, you collected samples over a long period of time, and sequence it, looking for specific mutations.
Specific single and specific mutation patterns are rather distinct between a “normal” infection, severe cases, and especially chronic, long-lasting infections in different settings (immunodeficiency being the easiest to research). It is also how the authors and researchers distinguish reinfections from persistent infections, although in extremely rare cases (I am aware of 5) an extinct lineage somehow resurfaces and reinfects someone.
Among the trends seen in the data, the one I am most interested in is the divergence between each variant and how many “persistent infections” it could theoretically cause, since each new variant had more immune evasive capabilities than the last one, and the data here reflects. Alpha, being among the most aggressive, had the least, while the “newer” Omicron lineage, more persistent infections were found.
Of note, 54 persistent infections spanned at least 56 days, with the longest infection lasting for over six months. Persistent infections yield mutations that are evolutionary beneficial to the virus in a distinct way, there is a trade-off during long-lasting infections were the virus trades off its incredible propensity to replicate, to survive inside the body (thus attaining a more sustainable replication rate).
Another important section is where the authors analyze and define persistent infections with viral rebounding. In rare cases even within persistently infected patients (in the context of the study a persistent infection is just an infection that lasts longer than the average) some of them experience a rebound, which the authors argue that reinfections within a short time are less likely given the context of their research, and the most likely mechanism is viral rebound. The virus would achieve this by persisting in the body at levels so small it avoids the detection threshold, a point I raised a few times.
Nonetheless, the observation of rebounding viral load dynamics in over 30% of cases is striking given that, in the absence of genetic information, they could have been misidentified as reinfections, depending on the definition used.
Individuals with a persistent infection report fewer symptoms later on the long-lasting infection than at the start of the sample collection, or sometimes remain completely asymptomatic, which is in line with my argument on the trade-off between replication vs persistence.
And lastly, a hot and controversial topic is the origins of Omicron variants. While no conclusive origin has been found for the Omicron variant, some argue it was a byproduct of a persistent infection in an immuno-compromised patient. Some immunocompromised patients experience persistent, months-long infection and they all share mutations seen in Omicron. Their paper is another piece of evidence for this line of argument.
Or, in a more conspiratorial flair, Omicron could have been serially passaged in immuno-compromised lab animals =). While I firmly believe given the evidence persistent infections are much more common than even the authors argue here, a counter-argument will be presented at the end.
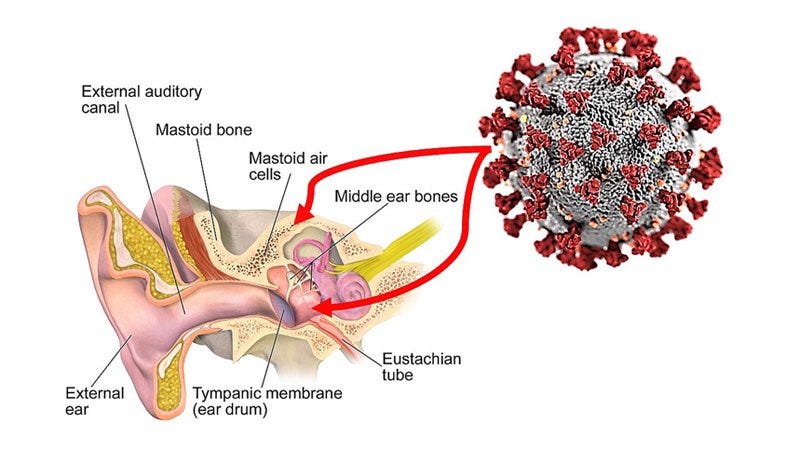
The silent reservoir? SARS-CoV-2 detection in the middle ear effusion of patients with Otitis media with effusion after omicron infection
Methods
This study, conducted from January to June 2023, spanned the Otolaryngology departments of two medical institutions in Eastern China. Patients manifesting OME subsequent to Omicron infection from both hospitals were subjected to comprehensive otolaryngological assessments, including pure-tone audiometry (PTA), tympanometry, otoscopic examination, and nasopharyngolaryngoscopy. Subsequently, MEE samples extracted from these patients were analyzed through RT-PCR to detect SARS-CoV-2.
Results
In this study, 23 patients (32–84 years; 57.5 ± 14.8 mean age; 47.8 % male) presented OME in 25 ears post-Omicron infection, with 21 (91.3 %) exhibiting unilateral symptoms. The median duration from infection to MEE sampling was 21 days (IQR: 25–46; range: 11–150). Predominantly, 64.0 % exhibited Type B tympanograms, and fluid accumulation was observed in 88.0 % of ears. SARS-CoV-2 was detected in 3 MEE samples (12.0 %), with cycle threshold values ranging between 25.65 and 33.30.
Conclusions
Our study highlights the potential effects of COVID-19 on the middle ear, suggesting a link between SARS-CoV-2 and OME onset. The virus, a significant contributor to OME, is detectable in the MEE nearly a month post-Omicron infection, indicating a potential alteration in OME treatment strategies and a risk of recurrence, emphasizing the necessity for otolaryngologist vigilance.
One of my arguments from 2023 (to my own detriment) is that SARS-CoV-2 persists in nerves, disagreement is fully understandable because pathogens persisting in nerves are rare and in their vast majority, not coronaviruses. The persistence of the virus or its pieces in the nervous system/nerves would explain a large amount of “novel” effects of the virus.
One of the most common forms of after-effects of Omicron infection, especially observed in China is Ottits Media Secretory, the build-up of fluids and “clogging” of the ear, especially the middle ear, and patients test positive for longer (thus the virus lives longer) in the middle ear than in the nasal cavity. The Middle Ear has a few nerves…
If you go back months and trace trends back, you can find similar, but unspoken trends in the West, when a Covid wave reaches peak and after, ear infections increase significantly from 2023, this one from a month ago, 2024.
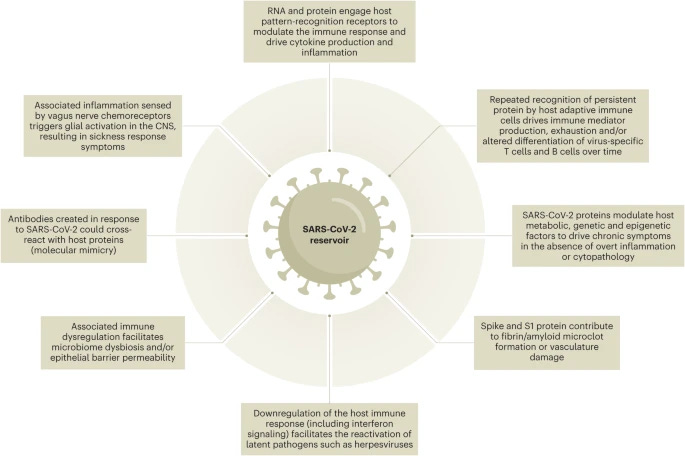
Before going further I would like to bring to your attention the following paper titled “SARS-CoV-2 reservoir in post-acute sequelae of COVID-19 (PASC)”. It is a great review that goes into many different aspects of the potential causes of Long Covid, it is worth your time and reading it.
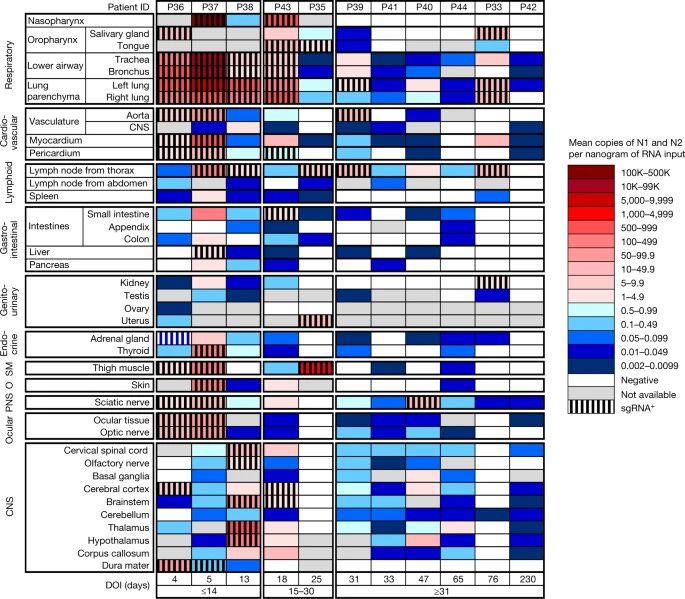
Autopsy and tissue biopsy studies have identified SARS-CoV-2 RNA and protein in a wide range of tissue types collected weeks or months after acute COVID-19 (refs. 24,25,26,27,28,29,30). Most of these studies were not designed to measure PASC symptoms, but nevertheless provide evidence that SARS-CoV-2 is capable of persistence in numerous reservoir sites (Table 1). One autopsy study identified SARS-CoV-2 RNA and protein in dozens of body tissues and brain obtained at least 31 d and up to 230 d after COVID-19 symptom onset31. Over 50% of these cases had persistent RNA in lymph nodes from the head and neck, and from the thorax, sciatic nerve, ocular tissue and in most sampled regions of the central nervous system (CNS) including the cervical spinal cord, brainstem and olfactory nerve. In one individual who died 230 d after mild COVID-19, SARS-CoV-2 RNA was identified in multiple anatomical sites, including several brain regions. Subgenomic RNA—a potential marker of recent viral replication—was identified in tissues after acute COVID-19, including in multiple tissues of a case at day 99—indicating that viral replication may occur in non-respiratory tissues for several months. Another study identified SARS-CoV-2 RNA in 80% of lung tissue samples obtained from individuals up to 174 d after COVID-19 onset32.
The paper goes to great lengths to explain the myriads of complex aspects of SARS-CoV-2 persistence, both the virus itself, its fragments, RNA, or reservoirs. The biggest “hurdle” in Long Covid research is the complex etiology, from my perspective, there isn’t one but multiple, going through the paper and my Index you will find a lot of correlations. From memory alone:
Persistence of fragments in cells, persistence in tissue
Potential reservoirs in organs (tissue), in the gut
A small percentage of patients will experience viral reservoirs inside cells
Potential persistence in nerves
Viral reservoirs in dental biofilms
Potential reservoirs inside biofilms elsewhere in the body
Reactivation of latent and chronic viruses, inducing immune dysfunction
Neurological sequelae, feeding into the cycle of gut inflammation
To understand how extensive SARS-CoV-2 infection and persistence can be, here is one of the most comprehensive papers on the subject.
SARS-CoV-2 infection and persistence in the human body and brain at autopsy
Coronavirus disease 2019 (COVID-19) is known to cause multi-organ dysfunction1,2,3 during acute infection with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), with some patients experiencing prolonged symptoms, termed post-acute sequelae of SARS-CoV-2 (refs. 4,5). However, the burden of infection outside the respiratory tract and time to viral clearance are not well characterized, particularly in the brain3,6,7,8,9,10,11,12,13,14. Here we carried out complete autopsies on 44 patients who died with COVID-19, with extensive sampling of the central nervous system in 11 of these patients, to map and quantify the distribution, replication and cell-type specificity of SARS-CoV-2 across the human body, including the brain, from acute infection to more than seven months following symptom onset. We show that SARS-CoV-2 is widely distributed, predominantly among patients who died with severe COVID-19, and that virus replication is present in multiple respiratory and non-respiratory tissues, including the brain, early in infection. Further, we detected persistent SARS-CoV-2 RNA in multiple anatomic sites, including throughout the brain, as late as 230 days following symptom onset in one case. Despite extensive distribution of SARS-CoV-2 RNA throughout the body, we observed little evidence of inflammation or direct viral cytopathology outside the respiratory tract. Our data indicate that in some patients SARS-CoV-2 can cause systemic infection and persist in the body for months.
Autopsies are quite rare in regard to Covid, and here the autopsies were done in unvaccinated individuals who died with Covid between April 2020 and March 2021, mostly older individuals.
The researchers found the virus not only in the lungs but in various other tissues of the body, including the brain, and in line with the evidence in some patients, the virus remained in the body for months. Widespread distribution throughout the body was often related to the severity of the infection with the earlier strains. The distinction between this paper and others is that they discovered this happens early in the infection in people who died.
Detection of SARS-CoV-2 RNA was observed in 84 distinct anatomical locations and body fluids, finding a higher viral burden (presence) in respiratory tissues compared to non-respiratory tissues during all stages of infection. They also found SARS in the heart, lymph nodes, gastrointestinal tract, adrenal glands, and eyes (especially earlier in the infection).
By tracking the decline levels of RNA in different tissues, they found that the viral RNA decreases quite fast in respiratory tissues, but it remains for a prolonged time in non-respiratory issues. Despite the persistence of the viral RNA across various tissues in the late stage of the infection, it was not found in plasma, but it was notable in the Central Nervous System, and once again in many areas of the brain. One of the patients who died still had the virus in the brain even after 230 days after symptom onset.
Despite the widespread presence of viral RNA, there was little evidence of inflammation or direct viral damage in tissues outside the respiratory system, suggesting that the virus can persist without causing noticeable tissue damage. In the 11 brains examined here, they found very few histopathologic changes, despite a significant amount of virus, but they found vascular congestion.
Vascular congestion (as the name implies, blood flows very slowly) is an unusual finding, and to this day has no known causes. However, it is a hallmark of an impaired blood-brain barrier and neuroinflammation. So a question left is what comes first. Viral infection and inflammation in other places in the body, lead to the compromise of the BBB, thus inducing vascular congestion, or the other way around.
And as the last paper, a very recent one, puts all this into perspective and adds both nuance and evidence to what is discussed here.
The persistence of SARS-CoV-2 in tissues and its association with long COVID symptoms: a cross-sectional cohort study in China
Unlike most studies conducted to date, especially on a large scale, this research focused on patients at different time points after recovering from mild COVID-19 (Omicron variant). While the evidence suggests that viral persistence is correlated with Long COVID, it remains inconclusive. The researchers aimed to understand both the presence of viral RNA and its association with Long COVID symptoms. The study was conducted at the China-Japan Friendship Hospital in Beijing, China, with a diverse group of patients undergoing gastroscopy, surgery, chemotherapy, or immunotherapy, or those hospitalized for other reasons, at 1, 2, and 4 months after infection.
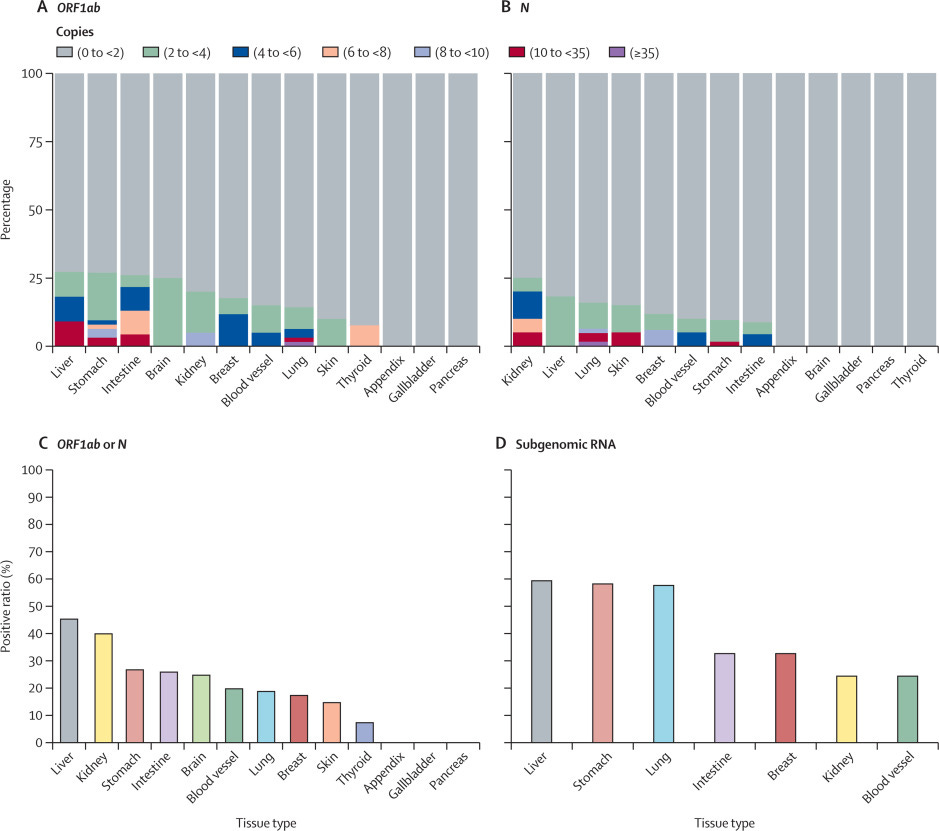
A total of 317 samples were collected from 215 patients. Tissue types from which the samples were collected included the lung, skin, intestine, blood vessel, kidney, breast, thyroid, liver, stomach, brain, pancreas, gallbladder, and appendix, as well as blood components (plasma, granulocytes, and PBMCs—lymphocytes, monocytes, natural killer cells (NK cells), and dendritic cells).
The researchers checked these samples for the presence of SARS-CoV-2 RNA at 1 month, 2 months, and 4 months post-infection. They found that 30% of the samples taken at one month, 27% at two months, and 11% at four months still contained viral RNA. This indicates that the virus can linger in the body long after the initial symptoms have subsided.
One of the most significant findings was that the virus persists for longer and can only be found in the blood and immune cells of immunocompromised patients, while other patients don’t experience the same persistence. Viral persistence at months 1 and 2 was the strongest indicator for the presence of at least one Long COVID symptom even at month 4, but viral persistence at month 4 was not indicative of Long COVID at that time point.
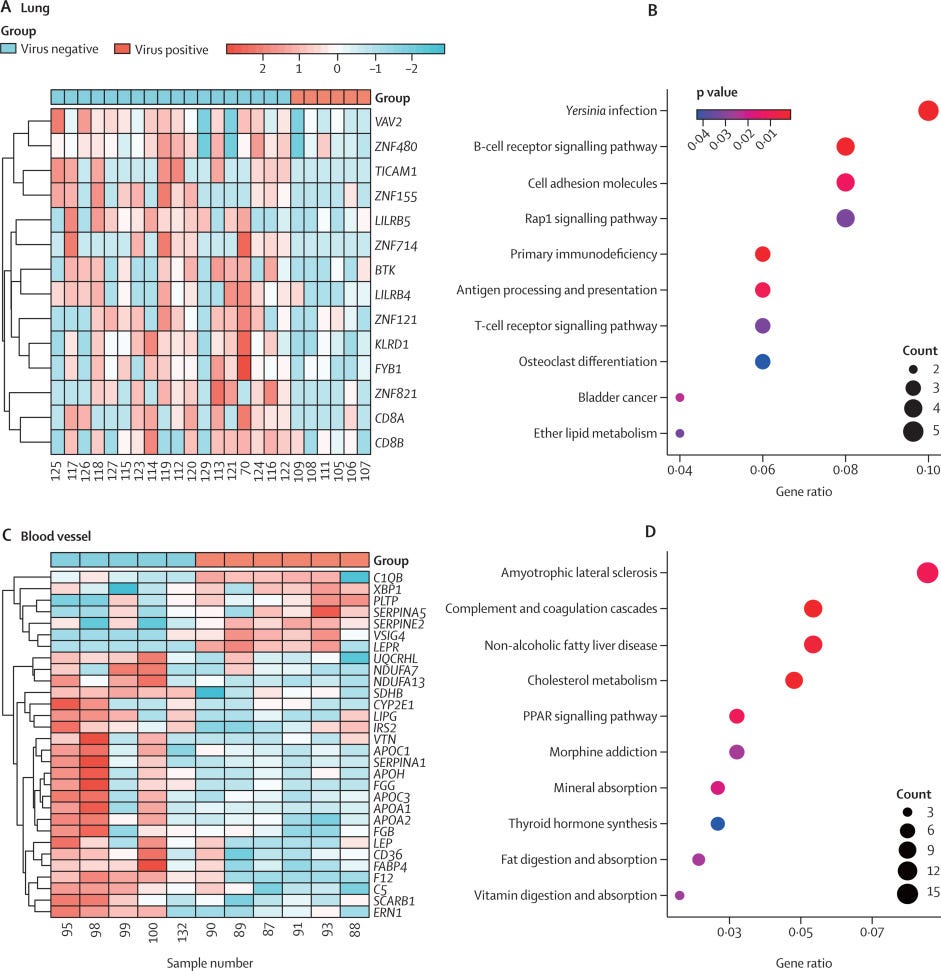
Further dividing these samples into three groups based on virus copy number, patients with higher viral loads (medium and high-virus groups) had a higher likelihood of developing Long COVID symptoms compared to the virus-free and low-virus groups. So, how does this happen? How does viral RNA persist for so long? To understand the potential mechanisms, the authors analyzed the genes in 24 lung tissue samples and 11 blood vessel samples.
In lung samples, innate and adaptive immune defense-related genes were found to be downregulated, as were zinc finger protein-related genes, which are important in defense against viral infections, especially SARS-CoV-2. In blood vessel samples, they observed dysregulation of genes involved in complement and coagulation cascades, as well as genes involved in cholesterol metabolism. This indicates that viral persistence itself affects cell function and cell metabolism.
The biggest problem with the persistence of viral RNA, even if we were to completely ignore the role of viral persistence at any level is that RNA and its fragments are bioactive molecules capable of affecting and influencing many biological processes. RNA fragments can trigger immune responses, and create inflammation, thus disrupting cellular and metabolic processes. How much of the unusual behavior of many pathogens in the last 2 to 3 years can be attributed not solely to the effects of the viral infection, but the persistence of viral RNA for extended periods of time ?
HMGB1 regulates Th17 cell differentiation and function, by regulating Th17 cell differentiation through HMGB1‐TLR4‐IL‐23‐RORγt pathway, then promotes IL‐17A production and aggravates inflammation process
Many of the genes analyzed by the authors play really complex roles in two things. Th17 differentiation and HMGB1 expression or direct direction. C1QB can interact with HMGB1 and produce a paradoxically potent anti-inflammatory effect. I suspect one of the myriad of reasons Metformin can positively impact Long Covid in many is precisely because it modulates the Th17 response and HMGB1 and shifts the body from a paradoxical immune response, towards equilibrium, helping the body clean itself from any viral remains.
Still, All roads lead to Rome.








After many months, I will be finally hiking at my favorite place this weekend. I still plan to write something else in the Fourth Option section, otherwise, any subsequent e-mail you get may as well be hiking pictures of nature in Brazil.
Do you take the BrainStack with you or let the system settle?
I was thinking of your advice last week when Joe Biden was up past his bedtime. They had him prepped and pumped the next day. I haven't seen enough of this pharmacology to have an opinion but I believe you and your fellows could have done a better job of priming him for two hours.
I have, however, seen a lot of "SunDowning" in patients in the dementia wards, and it may not be possible to counteract easily. Will the next "debate" be at lunchtime?
Fare well and freely, reply in a week or two if desired.
R