One Protein to Rule Them All
And in toxins bind them !
First and foremost, we need a few clarifications. The first and somewhat most important one is credit where credit is due. While I have mentioned and covered the protein we are about to talk about superficially, it never crossed my mind to further dig into it, after all, I live in a hamster wheel of my own making.
I chase dozens of pathways, with a few hundred interactions, which lead to more interactions, leading to more pathways. I will, quite often, miss “the obvious”. So sometimes I either have an eureka moment or a very random comment will set a cascade of cognitive events.
NatteringNaybob, a brilliant reader of mine was the reason I went down this nest. A little nudge sometimes is all it takes. He deserves the credit for this.
Secondly,
is the paramount expert on endotoxins, and endotoxins will be incredibly important here. Sometimes I describe him as “The Ralph Baric of endotoxin”. You should follow him and read his comprehensive work on all the forms of damage endotoxins can induce.I do not for a moment think I am the first person to mention this protein, but I suspect I will be the first to connect this many dots. If I could describe the protein with a meme, or a video, it would be with this.
As I shared a couple of times before, my mentor-friend and I, when creating our PAID hypothesis both agreed on a few variables, the most significant were:
The virus, and most precisely the Spike Protein must be flipping a single gene, or protein, quintessential for life, to be doing this much damage, at a systemic level, often paradoxically, creating dozens of cascades.
It also needs to be either occurring outside the cell and getting into the nucleus, or better yet, occurring directly at the nucleus and being transported outside. This would explain much of the “novelty” of the Spike-based damage.
The One Ring
High Mobility Group Box 1
HMGB1, also referred to as amphoterin, but mostly the amalgamation of letters is a protein encoded by the gene with the same name. High Mobility Groups are proteins that act at the nuclear level and help do many DNA-dependent things, like transcription, replication, recombination, and DNA repair. HMGB1 itself is among the most important proteins in humans, period.
HMGB1 acts in the nucleus of the cells but it can be “expelled” from it and act intracellularly and intracellularly, so it can do things in the nucleus of the cell, inside the cell, and outside the cell, this last one becoming something else, something…greater. In recent years HMGB1 has been considered a “sterile inflammatory protein”, the difference between sterile and non-sterile is simple.
Non-sterile inflammatory proteins are similar to all the complex letters and IL (Interleukins), IFNs (Interferons) we cover. Lego piece 1 needs to contact Lego piece 2, so an entirely different complete Lego comes and helps by releasing or breaking down other Legos. A sterile protein is different. If there is inflammation, the Legos start going, no need for 1 to contact 2, or all the complex interactions.
In simple terms, if there is inflammation or damage to a cell, HMGB1 is present, released, and acts and interacts with other proteins, it doesn’t need a complex set of interactions and signals to act. Like the One Ring influencing countless events indirectly.
2 years ago I wrote about the N-Terminal Domain of SARS-CoV-2 and its “uncharacteristic” similarity with human Galectin-3 (also a close analog to a piece of HIV), and it had so many interactions with important cells and functions, and so many effects I named it “chimeric protein”, and certain researchers describe as such. If the NTD-Gal3 is a chimera, HMGB1 is a hydra.
I recently wrote how Omicron can “eat away” your gut lining by a complex set of interactions, with one of the proteins being Galectin-9, thus causing bacteria and other toxins to “leak inside the body”, the other mechanism uses TIM-3 (Gal-9 will glue itself with TIM-3 and “kill” your immune cells, it can cause immune suppression too. Another TIM-3 ligand (glue itself and makes that protein work) is… HMGB1. Back in 2021, researchers were already connecting skin reactions in SARS-CoV-2 patients with the TIM3-HMGB1 axis.
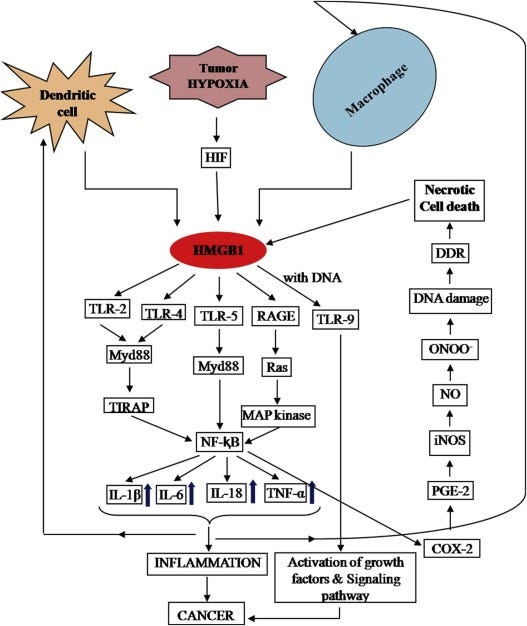
One of the receptors most engaged by the virus directly and indirectly and researched in regards to SARS-CoV-2 and Spike-mediated injury is TLR-4… In human cancer cells, when HMGB1 interacts with TLR-4 it induces the production of Galectin-9 and thus creates an immuno-suppressive environment. It makes it very hard for your immune cells to kill cancer. A common trend when reading untold amounts of HMGB1 papers is its dual-edge property, incidentally a trend that will be observed in this article.
In this regard, as a last line of defense, Galectin-9 decreased TNFα, IL-6, IL-10 and, high mobility group box 1 (HMGB1) and increased IL-15 and IL-17 increased Natural Killer cells numbers, Th17 cells thus protecting mice from polymicrobial sepsis. HMGB1 interaction can both be protective and damaging, even at the same time, a trend that will repeat itself in this article. Immunosuppression, TIM-3, and Gal-9 play a significant role in cancer, so our next step is that. Does HMGB1 play any role in cancer ?
The Shadows of Mordor
HMGB1 plays an important, and almost central role in tumorgenesis and progression, it has a dual role in cancer, but under the current circumstances it most likely will aid and accelerate the “bad” rather than help with the “good”. Blocking HMGB1 outside the cell can greatly help with cancer treatments.
HMGB1 involves different pathways associated with cancer development and progression.
HMGB1 inhibition can enhance response to immune checkpoint blockade in cancer.
HMGB1 is a novel and emergent therapeutic target for cancer prevention and treatment.
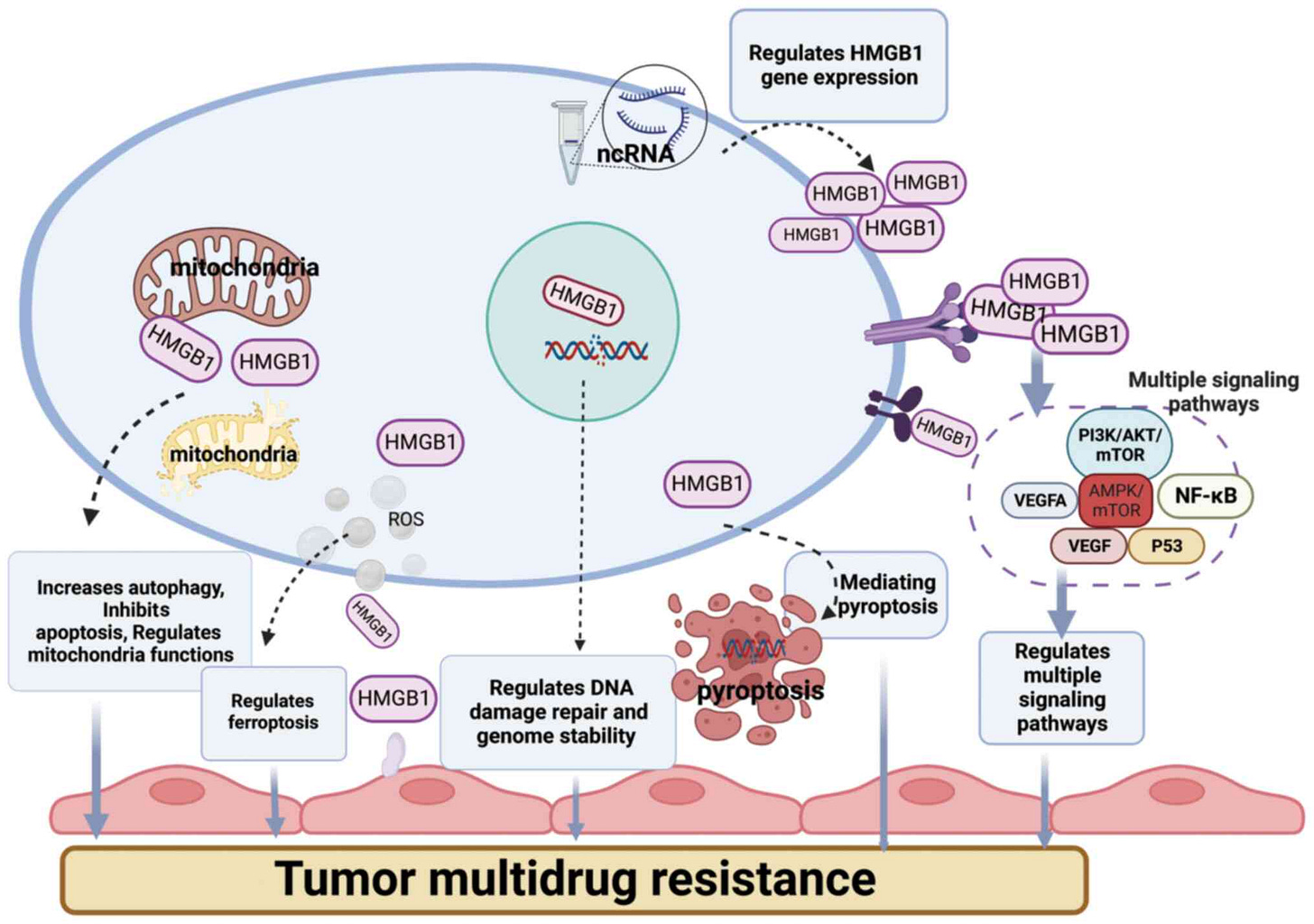
I recently wrote about how the Spike Protein S2 (the second part of the Spike) can inhibit P53, the “cancer protection gene” via a certain mechanism, so it is important to write that HMGB1 interacts directly with P53, this interaction inside the cell regulates the survival or death of cells in an intricate manner. HMGB1 and 2 have a cell-specific ability to down or upregulate P53 (and P73). It can regulate Depending on which receptor it binds, the place finds itself and cells, it can change multi-drug resistance in cancer. The graph below demonstrates partially the complexity of HMGB1.
Not only does HMGB1 play an almost central role in cancer, it mediates a myriad of different outcomes, but after a certain stage, it will most certainly “feed” the cancer, being subverted by the cancer cells to its own protection. Cancer and a state of immune suppression both at a systemic and cellular level go hand in hand. That is our next step.
If we are talking about the complex state of immunosuppression, Galectin, and TIM, we can’t forget Interferon Gamma. One of your body’s most potent “disease and cancer-fighting” cytokines so potent IFN-G itself can induce immunosuppression. IFN-G induces the release of HMGB1, and this is time-dependent (as with almost everything here). High mobility 1 mediated IFN-G modulation of Vascular Smooth Muscle Cells (the cells in your veins walls helping regulate blood pressure), in plain English, it creates and participates in a loop that narrows the veins.
If HMGB1 can participate and contribute to disease in your veins, inevitably the next line of inquiry would be the heart. It is an independent marker for all-cause mortality in patients with Dilated Cardiomyopathy. Its duality is highly complex in heart disease.
HMGB1 is a redox-sensitive molecule, so whenever oxidation either derived from cell or tissue injury happens, it will go through modifications, and at each stage, it can engage different receptors leading to the expression of different inflammatory proteins. In this regard, it plays a role in cardiac dysfunction, ischemic heart diseases, cardiac hypertrophy (thickening of the muscles in the heart) and cardiac fibrosis, myocarditis (via Th17 modulation), and is especially interesting for future reference diabetic cardiomyopathy
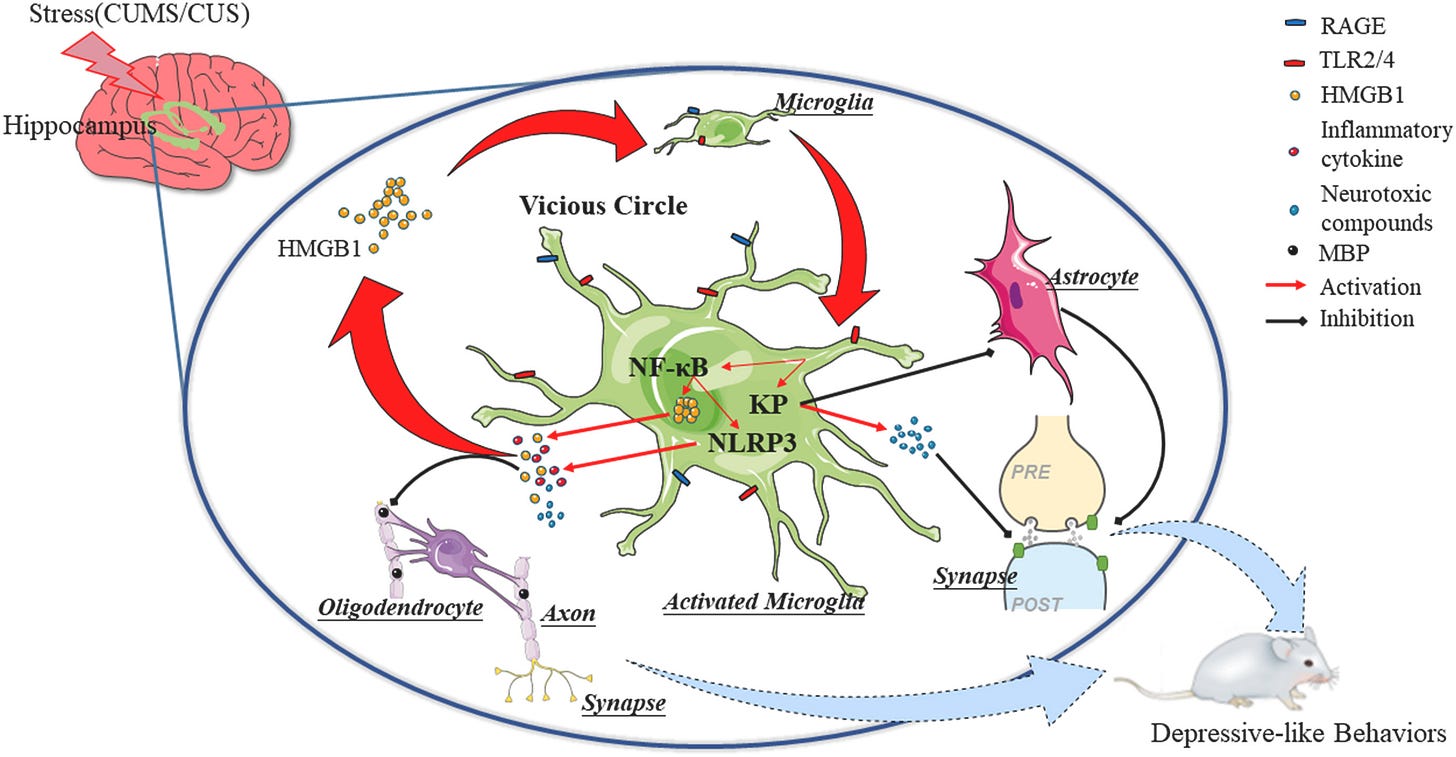
Going back to IFN-Gamma, if High Mobility participates in immunological loops the next logical step is wondering if this protein plays any direct or indirect role in one of the most important pathways, the Kynurenine Pathway.
“ds-HMGB1 induced depression in a manner associated with the kynurenine pathway, whereas oxidation of fr-HMGB1 evoked activation of the kynurenine pathway, resulting in depressive behavior.”
“In addition to HMGB1 and SIRT1, IDO, the rate-limiting enzyme of KP, was upregulated at the level of mRNA expression and enzyme activity in stressed hippocampi and LPS/HMGB1-treated hippocampal slices. The gene transcription of kynurenine monooxygenase (KMO) and kynureninase (KYNU) in the downstream of KP also increased both in vivo and in vitro.”
HMGB1 will activate the Kynurenine Pathway at different stages of its life cycle, and contribute to depression (depressive behavior), in addition, it is a primary part of the neuroinflammatory loop of chronic stress, leading once again, to depression. It can also contribute to anxiety.
“HMGB1 in nervous system diseases: A common biomarker and potential therapeutic target” is a great article to have a not overly complex perspective on the role of this protein in Parkinson’s disease, Stroke, Traumatic Brain Injury, Epilepsy, and Autism. HMGB1 acts via HMGB1/RAGE/TLR4 axis, and activation of TLR4 signaling leads to the upregulation of NADPH oxidase 2 (NOX-2)-dependent reactive oxygen species (ROS) production by immune cells, leading to persistent neuroinflammation. If you are paying attention so far, you raised your eyebrows a few times, because often we are reading RAGE and TLR4 as an actional axis for the One Ring.
What does the Kynurenine Pathway, the Heart, your veins and circulatory system, and all subjects covered so far have in common ? Besides Serotonin, they directly influence platelets, and thus…clots, or in “fancy scientific words” thrombotic-related diseases.
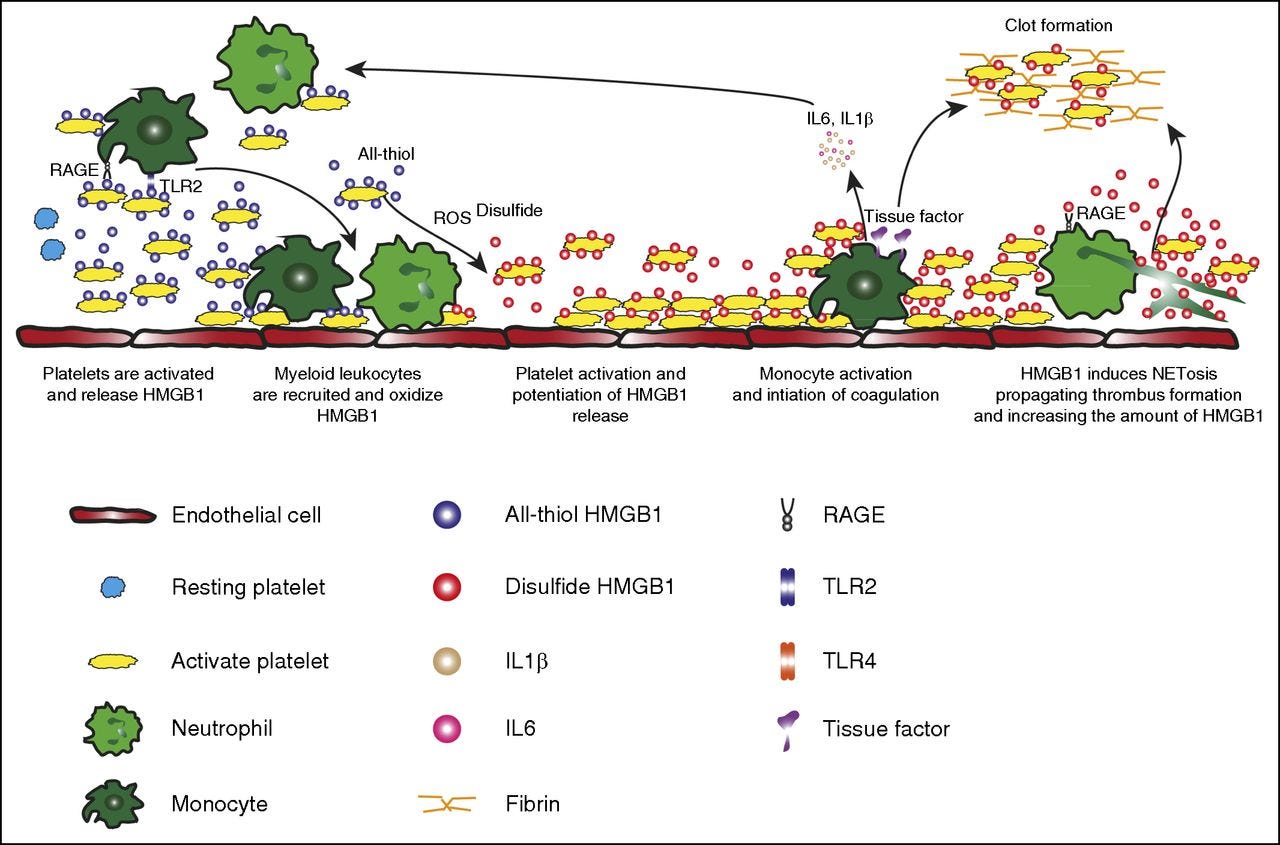
Mechanistically, HMGB1 directly participates in thrombosis or “clot formation” via numerous mechanisms, and as you can see in the image above, its levels are increased in many “clot-related events”. It can directly affect Nitric Oxide (NO) synthesis, especially in the endothelium, and it can directly inhibit fibrinolysis factors (how the body breaks down clots), to our interest here is the reference to HMGB1 and DIC, Disseminated Intravascular Coagulation, otherwise referred lately as “microclots”.
Other examples → “platelet-derived disulfide HMGB1 is a central mediator of the sterile inflammatory process in venous thrombosis”
“During sepsis, massive accumulation of HMGB1 in the systemic circulation would promote the development of DIC”. Sepsis is oh so very important to us.
Platelet-derived HMGB1 is one of the main mediators of thrombosis, via once again TLR4-dependent mechanisms, but not solely by, another mechanism and much more significant here is that platelet HMGB1 enhances the formation of NET (Neutrophil Extracellular Traps, a way for your body to trap pathogens and kill them, but it is also harmful to humans under specific circumstances) and specifically this clot effect is enhanced by extracellular DNA. Extracellular HMGB1 derived from activated platelets or NETosed neutrophils induces thrombosis and further aggravates NETosisc.
Practitioners of medicine will discuss what is more important, the heart or the brain, until the end of times, but since we already discussed the brain, the veins, and the nervous system, now we must see if HMGB1 plays any role in neurological conditions beyond depression and anxiety. The evidence so far already pointed out this.
HMGB1 induces microglial activation, aberrant synaptic pruning, and neuron dysfunction in an animal model of SAE (sepsis-associated encephalopathy), leading to cognitive impairment.
HMGB1 mediating C1Q adhesion on dendritic synapses of cultured cortical neurons through activated NMDA receptor, a recent second paper demonstrating the same
HMGB1-Mediated Senescence and Brain Inflammation Contributes to Cognitive Dysfunctions
Many of the complex mechanisms discussed so far occur in “life”, meaning cells are going through their complex set of interactions, but as the old anime catch-phrase goes “The goal of all life is Death”. And cells dying makes for an amazing way to release everything inside them, especially… HMGB1 (graph from The mechanism of HMGB1 secretion and release).

Now, up to this point, I cited only one paper from 2021 talking about SARS-CoV-2, and numerous others about the protein, and mechanisms of harm shared with SARS-CoV-2.
I could go on and on, and perhaps there will be a part 2 weeks from now, but I will end this in the next few paragraphs, here is what I want you to pay attention to. The first line in the graph. DNA damage, Ferroptosis, Cytokines and Infection, LPS, dsRNA, Pathogens, and just inflammation attracting neutrophils, all contribute to different forms of cell death, and inevitably the release of extracellular HMGB1. Extracellular is the most important one, because while HMGB1 is not like yeast, not “sticky”, it has peculiar and singular properties after certain interactions.
HMGB1 is potent and similar to a few of the proteins (IFN-Gamma, Gal-9), it has been a huge interest in being used as an adjuvant, especially as an improvement for vaccines, with potent anti-tumor effects. “Co-immunizing with HMGB1 enhances anti-tumor immunity of B7H3 vaccine in renal carcinoma”
Does mRNA vaccines raise the levels of HMGB1 ? Arguably the most important question here.
BNT162b2 induced damage-associated molecular pattern signals, including double-stranded DNA and HMGB1 peaking at 24 h after immunization
Hmgb1 was significantly increased in the mRNA-primed groups only compared to the negative control group
Long Covid ? The HMGB1 pathway influences the microbiome-immune axis, culminating in PASC
“While our step-wise pathway analysis suggests that Bifidobacterium modulates HMGB1 reciprocally, HMGB1 may also indirectly modulate Bifidobacterium abundance in PASC pathology”.
SARS-CoV-2 Infection Induces HMGB1 Secretion Through Post-Translational Modification and PANoptosis
Passive release of HMGB1 has been linked to various types of cell death; however, we demonstrated for the first time that PANoptosis, which integrates other cell death pathways, including pyroptosis, apoptosis, and necroptosis, is related to passive HMGB1 release during SARS-CoV-2 infection
The release and activity of HMGB1 in ferroptosis
Ferroptosis activators induce HMGB1 release.
Autophagy promotes HMGB1 release in ferroptosis.
Autophagy promotes HMGB1 acetylation in ferroptosis.
HMGB1-AGER pathway mediates inflammation response in ferroptosis.
One Ring To Rule Them All, and In Toxins Bind Them
As outlined at the start of this article, throughout the last 4 years my journey was to understand how a virus, and especially a chimeric Spike Protein could do so much, oftentimes paradoxically, subverting the entire immunological and cellular machine at multiple contradictory levels. At the end of year 2 (2021) I came to one conclusion, which was insanity at the time.
The virus or Spike Protein must interact with Endotoxins (LPS), and further yet it must interact with bacteria and biofilms at some level, it was the only mathematically possible explanation, regardless of how crazy it sounded in my head at the time.
The virus has a few nuclear localization sequences (amino acid sequences that enable it to bypass everything and get into the nucleus) but this can’t explain everything. There must be a way for the process of:
Spike inside a cell → gets outside the cell → picks up cargo → gets inside the cell again, whole or fragmented, and a molecular toxin-fueled IED goes off
HMGB1 is not sticky, but it interacts with many, many other proteins. In regards to endotoxemia (a lot of endotoxins in the blood) and sepsis, HMGB1 plays a leading role, leading HMGB1 to be given another name. “Endotoxin Delivery Protein HMGB1”.
“HMGB1 physically binds to extracellular LPS and subsequently utilizes a RAGE-dependent internalization process to deliver LPS into acidic lysosomes, where HMGB1 directly permeabilizes the phospholipid bilayer of the lysosomes.”
HMGB1 has 2 regions that can bind to LPS. HMGB1 physically interacts with LPS and targets its uptake into the lysosomes via the receptor for advanced glycation end-products (RAGE)11. HMGB1 in lysosomes permeabilizes the phospholipid bilayer in the acidic environment leading to the release of LPS into the cytosol and caspase-11 activation.
Interestingly enough this is one of the mechanisms by which SARS-CoV-2 will exacerbate disease severity, cause massive inflammation, and induce immunothrombosis (you can read this in the Endotoxin delivery article). The virus and especially the Spike Protein can subvert HMGB1, form new complexes with endotoxin and, well, you just read all it can do.
Endotoxin Tolerance, a state we can easily observe in the first few waves of vaccination, and now Omicron infection can NOT happen without HMGB1. Even the immuno-suppressive effects of the Spike are still mediated by the One Ring.
By this set of very complex interactions, the virus and especially its Spike Protein can bypass a myriad of cellular and immunological defenses, and deliver its cargo directly at the core of a cell, causing massive damage that “spills over”. LPS participates and can be found inducing or progressing almost if not all the conditions covered here. Endotoxins also mediate much of the damage from the Spike Protein.
To demonstrate even further how dynamic and multifunctional HMGB1 is, in the paper “The role of HMGB1 in COVID-19-induced cytokine storm and its potential therapeutic targets: A review” the authors propose, explaining in great detail, how could HMGB1 form RNA complexes with SARS-CoV-2 and induce cytokine storms. HMGB1 can bind to both DNA and RNA. HMGB1 (and 2) can also act as an Antimicrobial Peptide.
SARS-CoV-2 possesses specific fragments that act as xenoAMPs, forming complexes that enhance inflammation and are “hard to dissolve”. HMGB1 can bind to RNA, is also an AMP, binds to endotoxin… you can guess the rest =). So the last step here is biofilms. In my “hypothesis,” if we could call that, the manipulation of biofilms is essential, and a couple of years ago authors demonstrated that the Spike could interact with some, but not all.
From my perspective, for all this to work, the Spike Protein and virus would necessitate to actually manipulate biofilms.
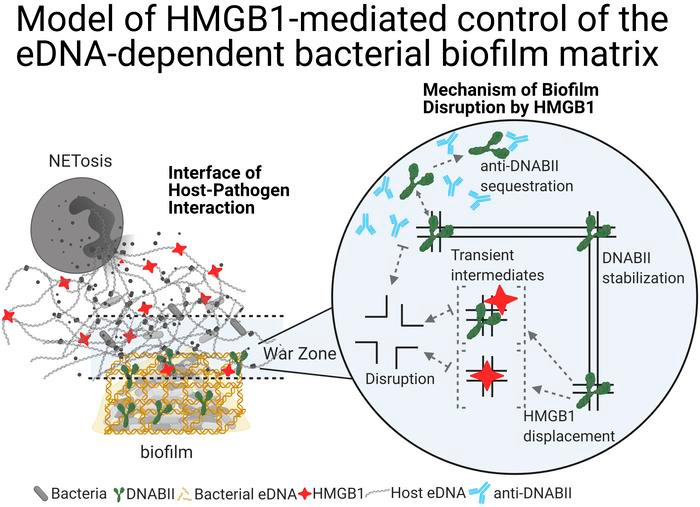
The extracellular innate-immune effector HMGB1 limits pathogenic bacterial biofilm proliferation
Herein, we describe an extracellular function of the vertebrate high-mobility group box 1 protein (HMGB1) in the proliferation of bacterial biofilms. Within host cells, HMGB1 functions as a DNA architectural protein, similar to the ubiquitous DNABII family of bacterial proteins; despite that, these proteins share no amino acid sequence identity. Extracellularly, HMGB1 induces a proinflammatory immune response, whereas the DNABII proteins stabilize the extracellular DNA-dependent matrix that maintains bacterial biofilms. We showed that when both proteins converged on extracellular DNA within bacterial biofilms, HMGB1, unlike the DNABII proteins, disrupted biofilms both in vitro (including the high-priority ESKAPEE pathogens) and in vivo in 2 distinct animal models, albeit with induction of a strong inflammatory response that we attenuated by a single engineered amino acid change. We propose a model where extracellular HMGB1 balances the degree of induced inflammation and biofilm containment without excessive release of biofilm-resident bacteria.
Your next thought should be “How could the vaccine-mediated injury without Spike toxicity” which does happen. It feels like magic, but it is just very advanced biochemistry.
…illustrated that HMGB1 was incorporated into sEVs through endosomal mechanisms. In conclusion, proinflammatory mediators released within sEVs can induce cell-specific inflammatory signaling in the brain without activating transmembrane receptors on other cells and causing overt inflammation.
Small Extracellular Vesicles (image the Amazon delivery system of your cells) can pick up and deliver HMGB1 without activating receptors, bypassing cell membranes, and causing inflammation. Stealthly. Given the extension and complexity, I will write the following in a shortly manner, because retroviruses and herpes viruses play a significant role in the long-term consequences of virus/spike damage.
Newcastle disease virus infection triggers HMGB1 release to promote the inflammatory response
Human Herpesvirus 6A Induces Dendritic Cell Death and HMGB1 Release without Virus Replication
The danger molecule HMGB1 cooperates with the NLRP3 inflammasome to sustain expression of the EBV lytic switch protein in Burkitt lymphoma cells
Waking up the Wringwraiths.
Now I must remain as the animated ad at the start of this article. Fast, agile, shape. How do you address HMGB1 ? You may laugh at the end.
The fires of 🌋 Mount Doom
β-Hydroxybutyrate as an epigenetic modifier: Underlying mechanisms and implications (yes, ketones will do it hehe)
Choline treatment suppresses HMGB1 release and improves survival in severe sepsis
The serum HMGB1 levels were significantly increased in RA patients in active phase. The serum levels of HMGB1 and inflammatory factors and VAS scores were decreased gradually with metformin treatment. HMGB1 might act as a novel therapeutic target for RA.
Aspirin delays mesothelioma growth by inhibiting HMGB1-mediated tumor progression
What a coincidence huh, my supplementation stack suggested in early 2021 addresses most of the injury mechanisms… huh. Well, never mind, all but coincidences.
Your support is welcome and I am grateful for those who choose to do it !








This took weeks to research, and dozens of hours to write by the simple fact, the more I wrote, the more I discovered, and I didn't even cover HALF of what I intended. So I will take the liberty to just chill the next 2 days.
Any meanigful post will only be published later next week, I will work on my short story for fun in the meantime. Any e-mail sent in the meantime will be "meaningless" from my perspective.
You will definitely need to read this one multiple times.
Have a fantastic weekend.
You have a very under valued substack mate, thank you.
While he's swinging everywhere and has a couple of oddities in his own past JJ Couey highlights some interesting co incidences in the names.
https://www.twitch.tv/gigaohmbiological?sr=a
Thanks again for your work mate and you're correct Geoff is a wealth of knowledge.