Lately, serendipity is finding ways in my life, events, and published papers with timely findings coming at one could say “the right time”. For the most part of my SARS-CoV-2 “deconstruction” and what basically one could entail as reverse engineering via meta-analysis and “intelligence” framework of the mRNA vaccine I had a few observations and arguments, that I consistently kept refining and contemporary research bringing evidence towards these observations.
Arguably, at this point, the whole SARS-CoV-2 part of my Substack are merely pieces of this hypothesis, perhaps one day in the near future I will attempt to solidify into one single “wall of text”.
One of the said arguments was that the mRNA vaccine has mostly a CD4 dominant response, it “obliterates” the CD8 response for months, and the mRNA also induces immunological imprinting and for the most part, made the vaccinated ignore the Nucleocapsid protein. And this mechanism was partially responsible for (at the moment) most of the adverse effects, now partly because it is much more complex, yet this mechanism of high CD4, low CD8, and no N immunity would drive a subset of people to develop Long Covid patients.
First, some snippets of things I wrote, with particular interest the first image.
For further analysis of diverse points, and quite a few more other substacks I wrote linked inside.
Nucleocapsid-specific T cell responses associate with control of SARS-CoV-2 in the upper airways before seroconversion
We find that soluble and transcriptional markers of systemic inflammation peak during the first week after symptom onset and correlate directly with upper airways viral loads (UA-VLs), whereas the contemporaneous frequencies of circulating viral nucleocapsid (NC)-specific CD4+ and CD8+ T cells correlate inversely with various inflammatory markers and UA-VLs. In addition, we show that high frequencies of activated CD4+ and CD8+ T cells are present in acutely infected nasopharyngeal tissue, many of which express genes encoding various effector molecules, such as cytotoxic proteins and IFN-γ. The presence of IFNG mRNA-expressing CD4+ and CD8+ T cells in the infected epithelium is further linked with common patterns of gene expression among virus-susceptible target cells and better local control of SARS-CoV-2. Collectively, these results identify an immune correlate of protection against SARS-CoV-2, which could inform the development of more effective vaccines to combat the acute and chronic illnesses attributable to COVID-19.
The abundant expression of internal viral proteins may therefore facilitate early antigen presentation at surface densities sufficient to trigger cognate T cells more rapidly than external viral proteins, leading to greater immune efficacy. This paradigm makes sense in the context of our study and cautions against vaccine strategies that immunize solely against the S protein of SARS-CoV-2.
Summarizing the paper above in simple but clear terms, SARS-CoV-2 control, of both the viral infection and its inflammatory responses occurs very early in the infection, in the very first start of the process, in the mucosal tissue of the nose. Something the mRNA completely bypasses, in fact, one of the early criticisms now backed by copious amounts of evidence was the fact that the mRNA doesn’t give you mucosal immunity and would fail to control the viral infection and spread. These findings are very pertinent to the subject and paper you are about to read.
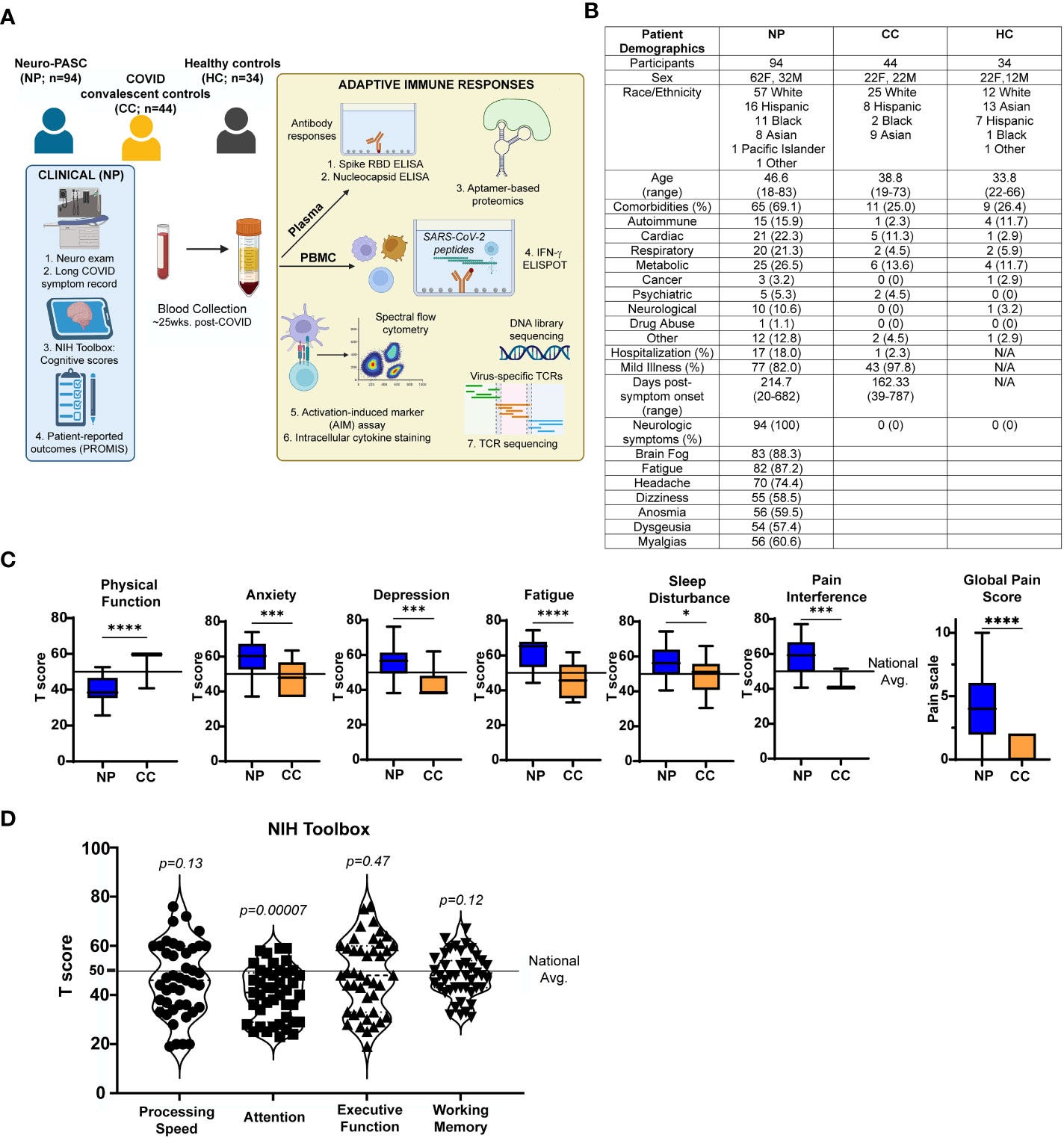
Before we go further, in case you are not aware, PASC (Long Covid patients) are not divided into sub-groups (I refer often to these as “subsets”), with each sub-group possessing specific, and sometimes overlapping, symptoms and markers, but often they are distinct enough in the markers territory so they can be classified as different types, Neuro-PASC being one of them. Scientifically they use 3 categories, I personally use more.
If you had the time or read the substacks linked or just consume a lot of scientific information in regards to SARS-CoV-2 you are very aware that T-Cell immunity is central to the defense and protection of your body against the viral infection. If your body, for a myriad number of reasons, fails to put up an effective and proper T-Cell response you get a severe acute infection that bears many negative short and long-term effects. Severely acute, and critically ill all had “defective” (sub-optimal, or impaired) responses to the virus, and besides all the physiological and immunological damage this entails, clear by all my recent substacks this entails secondary infections, both in and outside a hospital setting. This also entails more often than not, developing Post-acute sequelae of COVID-19 (PASC).
Alterations in T cell responses to SARS-CoV-2 nucleocapsid protein in Neuro-PASC
In this section of the study, researchers examined T-cell responses to different parts of the SARS-CoV-2 virus in individuals with neurological post-acute sequelae of COVID-19 (Neuro-PASC). They used a method called IFN-γ ELISPOT to measure these responses, Interferon Gamma being a very good marker to do this specific line of inquiry.
IFN-γ responses against S peptides were similar between Neuro-PASC patients and convalescent controls, but they found Neuro-PASC patients exhibited higher IFN-γ responses towards N and M peptides compared to the convalescent. To my surprise, the Receptor Binding Domain (RBD) was the same among all groups.
Furthermore, the Neuro-PASC patients displayed elevated levels of antibodies specifically targeting the N protein of the virus. They also showed increased activity in a type of T cell known as T follicular helper cells (Tfh), which play a role in antibody production. These findings suggest that the Neuro-PASC patients have heightened T cell and antibody responses directed at the N protein compared to individuals who have recovered from COVID-19. Importantly they found no antibodies against the (irrelevant on this content) Haemagglutinin protein (the H in HxNx) from Influenza viruses, so this was a SARS-CoV-2-specific response.
Enhanced IFN-γ production and CD4+ T cell activation to C-terminal region of N protein in Neuro-PASC
Having shown elevated IFN-γ responses towards the N protein in Neuro-PASC, the authors sought after to find if specific regions of the N protein enhanced the T-cell response and chose to focus solely on the N over the M because the N response is long-lasting and can persist up to 12 months post-infection, and most of their patients were enrolled in the study at the average of 7 months post-infection, thus assuring accurate detection. Since the early roll-out of mass SARS-CoV-2 vaccines in the area where they conducted this research they had to include both vaccinated and unvaccinated individuals (authors literally say it was very difficult to find unvaccinated after January 2021).
Nucleocapsid peptides were divided into 3 antigen pools, and elevated IFN-γ responses in Neuro-PASC patients were traced to the C-terminal region of the protein, particularly within amino acids 309-402. Furthermore, the IFN-γ ELISPOT data from the study participants aligned with these findings, indicating elevated T cell responses to the N protein in Neuro-PASC patients. Notably, the increased T cell reactivity in Neuro-PASC patients was predominantly attributed to CD4+ T cells.
Interestingly, there were no significant differences in HLA alleles (a set of genes that play a role in immune response) between the Neuro-PASC patients and the convalescent controls.
Attenuated N-specific CD8+ memory T cell activation in Neuro-PASC patients
In the section above the authors examined different T-cells subsets in both Neuro-PASC and convalescent. They found no differences between groups of most T Cells in an unstimulated state (meaning nothing activated them), therefore they went to investigate memory T Cell responses. The CD8 T-effector cells (TEM or TEMRA) that respond rapidly upon antigen re-encounter (re-infection), exhibited significant N-specific activation in convalescent but not in Neuro-PASC patients, suggesting a lack of T-cell memory responses in Neuro-PASC is detrimental to the condition.
A marked increase in CD8+ TEMRA cells was observed in Neuro-PASC, these are terminally differentiated subset types of CD8 cells, imagine “older and more experienced” T-Cells, these types of cells can accumulate during persistent viral infections and can contribute to immunosenescence, which refers to the gradual deterioration of the immune system related to aging but also to persistent long-term inflammation. This points towards an anergic state for these cells, meaning they have a reduced ability to respond to antigens.
On the other hand, CD8+ TEM from Neuro-PASC expressed higher levels of IL-6, one of the most potent inflammatory cytokines, in response to N antigens compared to convalescent controls. Per the words of the author themselves, IL-6 can play a regulatory role during viral infections by suppressing Th1 differentiation, thus promoting pathogen survival while exacerbating the disease, this was observed in SARS-CoV-1 infection, overexpression of IL-6 can lead to viral persistence and impair the development of CD8 T-cell memory.
I will now let the author’s own words end this.
Based on our data, it is, therefore, possible that enhanced IL-6 production from CD8+ T cells may upregulate KLRC1 and suppress CD8+ T cell function in Neuro-PASC patients, which may increase Neuro-PASC symptom severity. Together, these data illuminate a specific T cell signature composed of decreased CD8+ T cell memory responses and increased IL-6 stimulated by Nucleocapsid protein antigens that associate with Neuro-PASC.
KLRC1 , also known as NKG2A is a receptor expressed on Natural Killer (NK) cells and a subset of CD8 T cells, one of its main roles is suppressing immune responses, under chronic infections the abundance of KLRC1 on CD8 T-cells is associated with its anergic state, and often labeled as functionally exhausted, in simple terms, it has an impaired, weaker capacity to control infection.
Additionally, as we hypothesize that Neuro-PASC could be the result of a persistent or protracted infection, future studies would require testing of potential cryptic viral reservoirs, including stool or post-mortem multi-organ tissue sampling from Neuro-PASC patients.
“future studies would require testing of potential cryptic viral reservoirs” Do you mean, something like…biofilms perhaps ? Besides being facetious I could propose another point I raised, the way the body will deal with the N protein when not properly addressed would be by building complex protein structures around it.
Since this substack is already rather long and complex, I will write tomorrow about how one may address these problems in short form, adding the potential for a novel treatment too, that may be beneficial for many PASC patients. It is quite a remarkable paper too.
Recommendation: Treatment of clinical long COVID encephalopathies with nasal administered mesenchymal stromal cell extracellular vesicles
If you choose to support this work in whatever form, thank you !





Thanks for Sharing much appreciated 👍
Have you written about IL-16 upregulation secreted by CD8 cells in relation to Heart or other organ Damage? Looks like miR-146a might be relevant. Provides a link to Endotoxin in jabs.