SARS-CoV-2 and the Gut
Part I of I don't know how many
I hope you are all having a good weekend, I postponed this post because I was deciding on length. And decide to minimize it to directly related papers, and not dive deep into everything they entail, I will in the future, but Covid, gut, and microbiome are an extensive (sometimes controversial) subject. To start.

Gastrointestinal symptoms and fecal shedding of SARS-CoV-2 RNA suggest prolonged gastrointestinal infection
Highlights
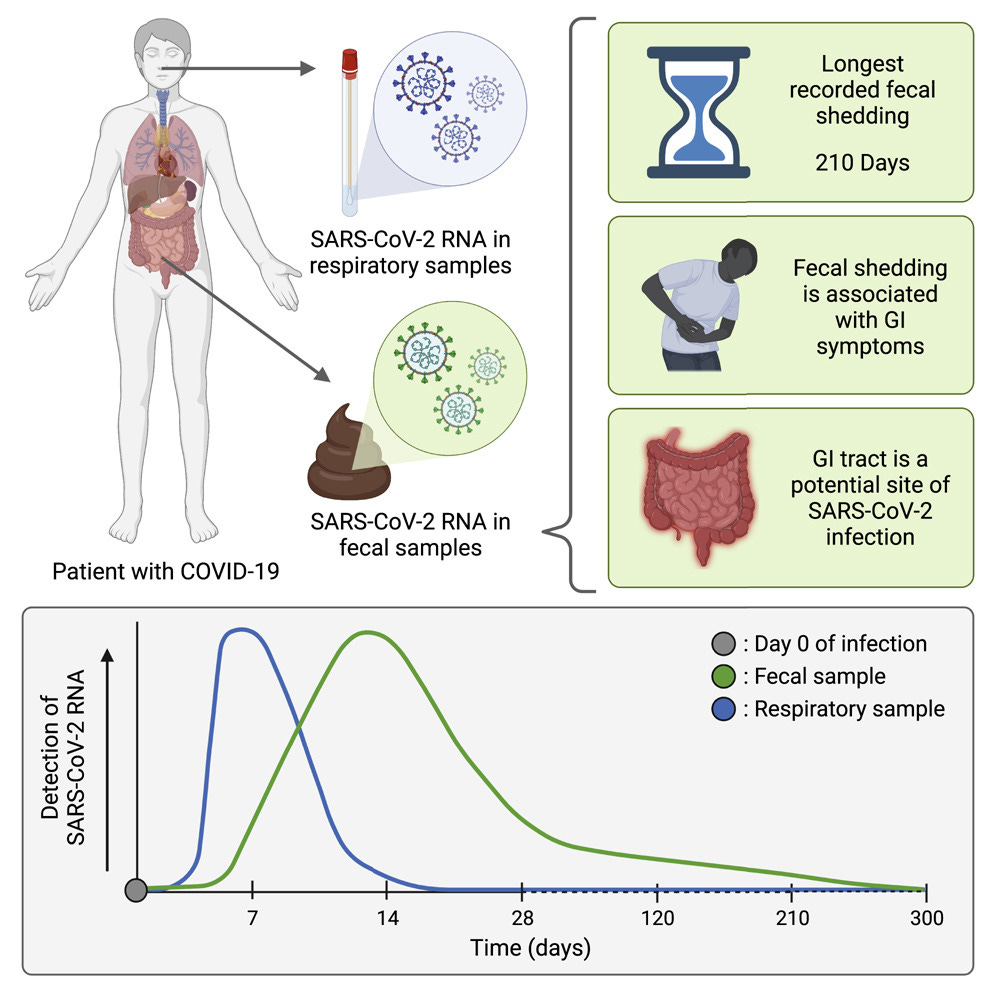
Approximately one-half of COVID-19 patients shed fecal RNA in the week after diagnosis
Four percent of patients with COVID-19 shed fecal viral RNA 7 months after diagnosis
Presence of fecal SARS-CoV-2 RNA is associated with gastrointestinal symptoms
SARS-CoV-2 likely infects gastrointestinal tissue
The reader should be aware that this discussion is as old as the pandemic itself, ever since late January 2020 there were discussions on the gut and SARS-COV-2. In case you are unaware the digestive tract and gut epithelial cells have high expression of ACE2.
Findings
Fecal SARS-CoV-2 RNA is detected in 49.2% [95% confidence interval, 38.2%–60.3%] of participants within the first week after diagnosis. Whereas there was no ongoing oropharyngeal SARS-CoV-2 RNA shedding in subjects at 4 months, 12.7% [8.5%–18.4%] of participants continued to shed SARS-CoV-2 RNA in the feces at 4 months after diagnosis and 3.8% [2.0%–7.3%] shed at 7 months. Finally, we found that GI symptoms (abdominal pain, nausea, vomiting) are associated with fecal shedding of SARS-CoV-2 RNA.
Conclusions
The extended presence of viral RNA in feces, but not in respiratory samples, along with the association of fecal viral RNA shedding with GI symptoms suggest that SARS-CoV-2 infects the GI tract and that this infection can be prolonged in a subset of individuals with COVID-19.
Interestingly, in the few studies that have investigated longitudinal fecal samples, prolonged fecal shedding of SARS-CoV-2 RNA can occur even after respiratory shedding ceases. Indeed, in one notable pediatric case, fecal viral RNA shedding extended beyond 70 days after disease onset.
Worth noting, as usual, that these dynamics are not systemic, specific subsets of people experience different long-term effects (I often refer to these as infection after effects) of the viral infection, these dynamics somewhat changed with Omicron FYI, now how these will change further with new subvariants only time will tell.
I do not think there should be an actual debate IF SARS-CoV-2 persists in the gut, but how. I wrote about it a few weeks ago about how after my Omicron infection, I stopped everything I was taking, so the virus would persist, I got long term GI symptoms, so I could find a way to “get rid” of the virus in the GI. The post below is where I referred to this.
Spoiler alert, this is one of the GI mechanisms I firmly believe SARS-CoV-2 uses.
If SARS-CoV-2 RNA shedding in the feces is indicative of a GI infection, this suggests that SARS-CoV-2 infection of the GI tract can continue after clearance from the respiratory tract.
Given the post below, here are some sources. From this paper.
Biofilms provide a protective shell for pathogenic bacteria to evade host defense (Tytgat et al., 2019). They are an ideal environment for pathogenic bacteria to build virulence, so the occurrence of some mature biofilms on healthy tissues may be an early warning signal for the transition to a damaged gut.
The gut is also the most invasive site for many bacterial and viral pathogens (Palmer et al., 2007). Despite direct exposure to a large number of microorganisms and foreign antigens, a unique intestinal mucosal immune system maintains the homeostasis of the gut.
There is constant interaction between the epithelial cells and the gut microbiota, both of which have been implicated in the regulation of intestinal barrier function (Natividad and Verdu, 2013). The commensal microbiota is able to shape the intestinal barrier structure by controlling physiological paracellular permeability and enhancing the mucus layer (Hayes et al., 2018).
The paragraphs above are but a small sample of the extensive literature on biofilms, the gut, and everything linked to it.
SARS-CoV-2 presence in the GI tract has additional relevance to patient health. The GI tract is a highly immunoactive tissue, and SARS-CoV-2 antigens in this body site may hone a humoral immune response against variants of the SARS-CoV-2 virus. Furthermore, prolonged presence of SARS-CoV-2 in the GI tissue may also have an impact on the hitherto mysterious phenomenon of post-acute sequelae of SARS-CoV-2 infection (PASC) or “Long COVID,” where individuals suffer from an unusual constellation of symptoms even after recovery from the respiratory SARS-CoV-2 infection. Taken together, it is critical that we understand whether or not the GI tract is infected and the dynamics of the infection in this tissue, from the standpoint of both the acute infection and the long-term sequelae of COVID-19.
Here, we sought to better define the features of SARS-CoV-2 presence in the GI tract and its relevance for short- and long-term human health.
If the reader doesn’t know by now, up to 50% of your neurotransmitters are made in your gut, it is a neuromodulator, a (literal) second brain, and an immunomodulator, among other things, the gut can literally shift your entire immune response and creates up and down enough to long-term low-grande inflammation gives a propicious environment for the setting in of chronic infections and diseases. This was always the basis of my argument for Chronic Covid (which I was among the first to propose, mentioning not for the sake of being first, but to prove a point that I actually understand the dynamics and pathways of the virus and the non-linear dynamics attached to this).
It is safe to deduce, from all the evidence you can research that whatever happens in your gut, will also reflect on your brain. If we take into account the bacteriophage aspect of the virus, and the predilection of the virus to hitch a hide in the nerves, it could be easy to link both of these to the vagus nerve, a dysfunction far too common among a subset of Long Covid patients.
The authors go to describe their data, the proteins they chose to measure, and how long the “shedding” lasted. A point I didn’t see many on social media raise.
Among the 23 fecal samples collected at 10 months (300 days), none were positive for SARS-CoV-2 RNA. It should be noted that the presence of viral RNA in the feces at the later time points could be the consequence of prolonged infection and viral RNA shedding or the consequence of a re-infection.
Finally, it would be interesting to sequence fecal viral RNA from participants with extended shedding to evaluate the persistence of the original virus variant, evolution of the original variant, and/or potential re-infection by the same or a different SARS-CoV-2 variant. Unfortunately, one of the limitations of current technologies for sequencing variants from complex matrices such as stool is the requirement of an adequate concentration of virus to be able to either amplify or assemble the virus from direct or enriched sequencing.
Of note, this study was carried out prior to the emergence of the strains (Omicron, Delta) that are prevalent today. Different strains may have different relative tropisms to the respiratory versus GI tract and may exhibit differences in clearance rates. This may be the consequence of their inherent biology as well as the immune status of the host due to underlying disorders, prior COVID-19 disease, and natural immunization, or vaccination.
This paper was mostly done with Alpha infected patients, a variant known not only for being hyper-aggressive but also one that caused GI symptoms in many millions globally, also a variant known to cause secondary bacterial infections like nothing else (well, besides the vaccines that use Alpha spike…). Delta didn’t have GI tropism to the extent of Alpha, but by my own experience, and you can easily find hundreds of tweets about it on social media, Omicron now does, and for long periods of time.
Given the vaccination, its effects have on the immune system, and how many of the vaccinated fail to clear the viral proteins (not the virus, the “viral trash” after your body deals with the living pathogen, which is one of the causes of the low-grade inflammation, as I often write about it here), it is safe to assume this will be a rather complex and difficult study to execute, many of the vaccinated are graced with a reinfection every quarter, and other don’t, how do you separate those, among other variables is the question.
If you want a very simplistic version of how that is happening and how it entails to everything else, see this post that touches the surface of amyloidogenesis, and how viral fragments can persist inside the body (I will refer to this post in my Amyloid post too, it is super short).
This paper also had a section about the authors using Interferon Lamba in a randomized setting to see the effects it had on the shedding, and after initial benefit, none. This would be fairly easy to deduce since Interferon Lamba often has an anti-inflammatory effect. It would be a good data point for interdisciplinary research if they measure inflammatory markers in the cohort (group of people) in this study.
Now to a paper about the Gut and Long Covid (often referred in papers as PACS, Post Acute Covid Syndrome).
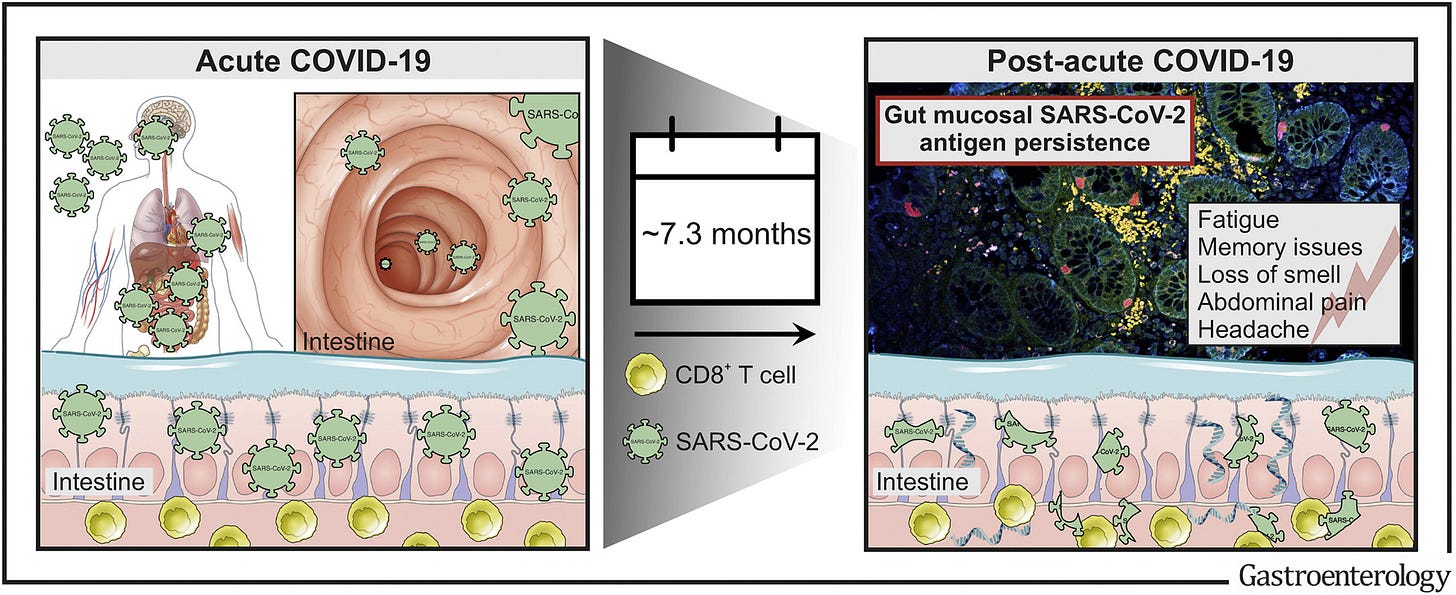
Postacute COVID-19 is Characterized by Gut Viral Antigen Persistence in Inflammatory Bowel Diseases
Results
We report expression of SARS-CoV-2 RNA in the gut mucosa ∼7 months after mild acute COVID-19 in 32 of 46 patients with IBD. Viral nucleocapsid protein persisted in 24 of 46 patients in gut epithelium and CD8+ T cells. Expression of SARS-CoV-2 antigens was not detectable in stool and viral antigen persistence was unrelated to severity of acute COVID-19, immunosuppressive therapy, and gut inflammation. We were unable to culture SARS-CoV-2 from gut tissue of patients with viral antigen persistence. Postacute sequelae of COVID-19 were reported from the majority of patients with viral antigen persistence, but not from patients without viral antigen persistence.
The author of this paper decided to measure the presence of the Nucleocapsid protein to test the persistence of symptoms, you might ask why, and it is simple, a decent body of literature about PASC points out the N protein being one of the origins of the long-term dysfunction presented in many of these individuals. And for once, vaccination status ain’t solely to blame. Since the Gut is a very important part of your physiology, it would be no different with IBS, and its known aspect of immune dysfunction.
What many of the PASC afflicted population have in common is immune dysfunction, clinical or subclinical, the viral infection will accelerate it. Especially mitochondrial dysfunction (yes, it mostly boils down to mitochondrial dysfunction and cascade from there.
It has been known for a while that SARS-CoV-2 infects (DOES NOT REPLICATE, IT IS NOT AIDS) T Lymphocytes. Coincidentally enough, it also talks about the N protein, and both of these mechanisms work together.
Why I am referring to all of this ? Well, you add these different mechanics and dynamics and you get the persistence of viral fragments in cells and in the gut. Without a proper immune response, which people afflicted by IBS often suffer from, add the mitochondrial dysfunction, the death of T Lymphocytes, and the persistence of viral fragments creating local low-grade inflammation, you get the paper above.
I made an observation in mid-2020 where I stated SARS-CoV-2 will attack you where you are weakest, so I find it rather interesting that each subset of patients in this study was affected in different parts of the gut. Omicron hit me hard in the colon, for example.
Results
SARS-CoV-2 Antigen Persistence Frequently Occurs in the Gut Mucosa
Patients recruited in this study had acute COVID-19 (PCR-confirmed SARS-CoV-2 infection) 219 days (range, 94–257) before endoscopy, which was performed to evaluate disease activity of an established IBD (ie, we did not select for patients with postacute COVID-19). At endoscopy, patients were evaluated for postacute COVID-19 symptoms (using a questionnaire) and viral antigen persistence in the gut. We included 46 patients with characteristics, comorbidities, risk factors, and medical history summarized in , and COVID-19–related clinical and biochemical characteristics shown in. Briefly, 91% (42/46) of patients had experienced mild acute COVID-19, and 45% (21/46) of patients reported at least 1 postacute COVID-19 symptom. On the day of endoscopic examination, all patients provided a negative COVID-19 nasal or pharyngeal PCR test and did not display clinical signs of a respiratory infection. Endoscopy revealed that the majority of patients with IBD (59%) were in remission. We evaluated viral antigen persistence by analysing biopsy specimens from the small and large intestine with quantitative polymerase chain reaction (qPCR), immunofluorescence, and viral culture from gut tissue. Moreover, we evaluated a systemic SARS CoV-2–directed immune response and gut inflammation (indicated by fecal calprotectin; ). Notably, 70% (32/46) of patients displayed a positive qPCR signal in at least 1 segment of the gut (ie, duodenum, ileum, or colon) . We detected viral RNA in 31% of biopsy specimens, with expression of the RdRP in 13.6% of biopsy specimens, the surface glycoprotein (Spike) in 11.4% of biopsy specimens, the nucleocapsid phosphoprotein (Nucleocapsid) in 10.6% of biopsy specimens, and the envelope protein in 6.1% of biopsy specimens . Detection using qPCR was unrelated to the intestinal location, the time from COVID-19 diagnosis to endoscopy, and IBD activity . SARS-CoV-2 expression was not detectable in stool from patients in this cohort).
The more acute/severe your infection, the more “persistent” and long-term after-effects you might experience, here they explain how they went to test their hypothesis, which is good, but I personally have problems with it, relying on PCR, but they used ELISA, standard among research. A lot of science talk to say they found viral fragments (that is what antigens are, viral fragments that elicit an immune response from your immune system). The following part caught my eye.
Nucleocapsid immunoreactivity specifically localized to epithelial cells and possibly stem cells , and to a lesser extent to CD8+ T cells in the epithelium and lamina propria.
A paper very few discussed was the one I discussed here on how SARS-CoV-2 spike protein damages stem cells.
Finally, we sought to define potential mechanisms of viral antigen persistence in the intestine.
We analyzed SARS-CoV-2–associated humoral and cellular immune responses with ELISAs, IGRA, and by surface and ICFC of peripheral blood cells (using peptide pools mapping the spike and the nucleocapsid proteins). Blood-derived immune cells from patients with gut antigen persistence exhibited a comparable IFN-γ release on SARS-CoV-2 nucleocapsid exposure as patients without antigen persistence . We rather noted that patients with gut viral antigen persistence more frequently lacked evidence of antinucleocapsid IgG antibodies and that anti-TNF immunosuppressive therapy was associated with impaired inflammatory T-cell responses on nucleocapsid peptide stimulation.
And here we have it, patients with gut antigen (viral fragment) persistence exhibited the same antiviral immune response as people without it, the difference was that patients with that persistence had no anti-N protein antibodies, and were often followed by anti-TNF (Tumor Necrosis Factor) therapy is associated with the lack of this response, and as I demonstrated earlier, other pathologies themselves will do this too.
We argue that viral antigen persistence reflects incomplete clearance of SARS-CoV-2 rather than subclinical (latent or persistent) infection because we were unable to replicate virus from biopsy-derived tissue. In line with this, we usually detected only some (but not all) viral transcripts in biopsy specimens from the same patient. Our experimental data rather suggest that immunosuppressive therapy with or without genetic predisposition (affecting the immune system) may promote incomplete viral clearance.
Indeed, we find that some patients exhibit a lack of humoral nucleocapsid IgG antibodies, which is pronounced in those with gut antigen persistence. In line with this notion, a previous report demonstrated immune dysregulation, eg, a decrease of nucleocapsid-specific IFN-γ–producing CD8+ T cells in patients with postacute COVID-19.
This quote explains itself. And for the record, as I earlier made it clear, I do believe in some people SARS-CoV-2 will not only replicate in the gut after becoming latent, but it will interact with the bacteria, but that would be in a small subset of the entire global population.
I agree with the conclusion by the authors, well, I argued the same for months, incompetence to clear the virus due to immune dysfunction, among other mechanisms is what is driving a lot of the long-term damage we are witnessing. The viral infections aided by the immune dysfunction inflicted by the vaccines are slowly chipping away at people’s immune systems, and if they don’t take proper care, it will stack. Some reach a tipping point earlier than others, and some are never affected at all (My own mother is one that literally laughs at the viral infections of all sorts including all SARS-CoV-2 strains, and she is not healthy…lol).
The last bold part is the most important one so far, by the fact that this is precisely what all the vaccines do NOT give you, a strong, IFN-γ–producing CD8+ T cells response. Per my recent Omicron posts, breakthrough infection will “seroconvert” you to it, at what rate nobody knows.
In the end, the persistence of viral fragments in your gut is a problem not because of the preposterous bullshit some spew, but rather by the continuous shifting of your immune system, and eliciting a systemic immune response that is often inflammatory, plus my own perspective on the bacterial side of this equation, which leads credence to a chronic state of infection of all sorts.
Both of these papers could be linked to the mechanisms discussed here.
If you are anxious, if you don’t suffer from autoimmune issues, solving this viral persistence is rather “simple”. Adding specific fiber types to your diet (Wheat bran was what I used), enough antioxidants, especially the ones that aid the gut as a byproduct (NAC is the main one here, Vitamin C is also a good one!!), and probiotics will help with these. When I tested it on myself, it took 4.5 weeks to completely “clear” whatever Omicron did to my gut, but remember, I purposefully let it “run its course”. The majority of the population won’t have this problem, and if you take any sort of supplements, the chances are .1% (that is well below 1%, there is a point there).
As a final note, I recommend you read the following paper, which is a fairly interesting read. Both the posts that I linked here are very good reading to having a broader understanding of what is going on, in my opinion, the Stem cells is the most important one not talked about.
Potential intestinal infection and faecal-oral transmission of human coronaviruses
I hope everyone has a very nice Sunday, probably be no posts tomorrow.




ITS ALL ABOUT THE MITOCHONDRIA BABY !
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7998235/
Among many other papers. The Brain post has the first of what an answer could look like. SARS-CoV-2 hits your mitochondrial function and run (virus dies), and something else hits you, and damage adds up.
Have a nice weekend people =D !
Here is a clinical case, pay attention to the degree of infection, it is always acute/severe. I do rather agree with the authors of the second paper, incompetent clearance.
Prolonged fecal shedding of SARS-CoV-2 in a young immunocompetent COVID-19 patient: A case report and literature overview
https://onlinelibrary.wiley.com/doi/10.1002/jmv.27694