I will try very hard to attempt to simplify a very complex topic, this is merely a fraction of this specific pathway too. And for full disclosure, I wouldn’t be fully aware of this pathway if not for Theo on Twitter, a remarkable brilliant individual, he was pretty much the first person I ever saw researching and uncovering huge amounts of information on this specific pathway, which I subsequentially named “Theo’s pathway”. Credit where credit is due.
First, we need an overview of what are integrins, and if after reading this substack you find yourself wanting to delve deeper into the subject, the first referenced paper is a fairly extensive and amazing read on the topic.
Targeting integrin pathways: mechanisms and advances in therapy
Integrins are considered the main cell-adhesion transmembrane receptors that play multifaceted roles as extracellular matrix (ECM)-cytoskeletal linkers and transducers in biochemical and mechanical signals between cells and their environment in a wide range of states in health and diseases. Integrin functions are dependable on a delicate balance between active and inactive status via multiple mechanisms, including protein-protein interactions, conformational changes, and trafficking.
In a simplified manner, integrins are a superfamily (an academic word when a protein has a lot of “members”) that are responsible for cell adhesion for many proteins outside the cell, they are located on the cell surface and their response is very contextual and situation-dependent.
Beyond classical ECM mediators, integrins are also reported to interact with a diversity of non-ECM proteins on the surfaces of prokaryotic, eukaryotic, and fungal cells, as well as a range of viruses.15,16 In addition, integrins can also be exploited as cell-surface receptors for growth factors, hormones, and polyphenols.17
There are 3 “classical” ways for proteins to interact with Integrin, but our interest is lies in two of them.
RGD-binding integrins
RGD-binding integrins refer to a class of integrins that bind with the tripeptide motif Arg–Gly–Asp in ECM proteins, including αvβ1, αvβ3, αvβ5, αvβ6, αvβ8, α5β1, α8β1, and αIIbβ350,51.
Leukocyte cell-adhesion integrins
Leukocytes constitutively express several types of integrins, including α4β1, α9β1, αLβ2, αMβ2, αXβ2, αDβ2, α4β7, and αEβ785 (Fig. 3). Among them, integrins containing the β2 subunit are most abundant in leukocytes; therefore, integrin β2 is also called a leukocyte integrin.86
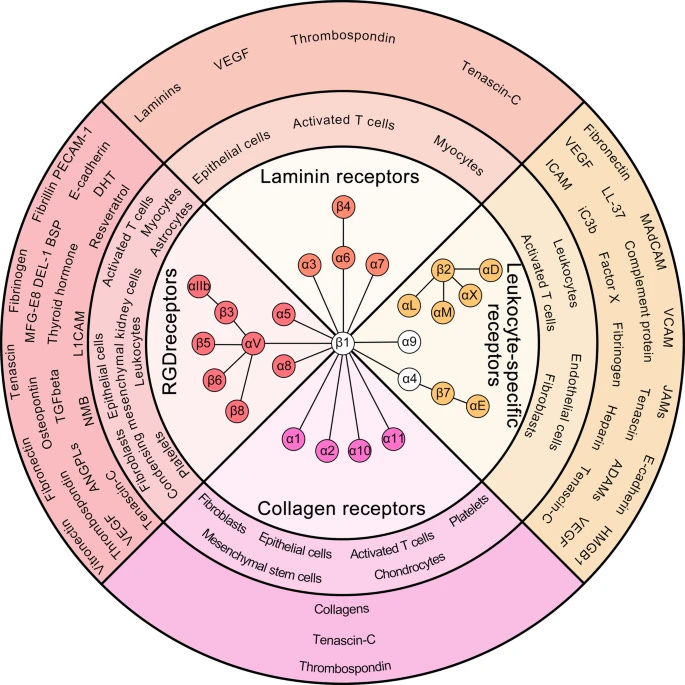
Per the image above, each group of integrins is responsible to interact with a myriad of proteins and exert extensive and complex functions. αLβ2 is involved in enhancing the phagocytosis of bacteria by neutrophils, less presence of it means less capacity to get rid of and clean bacterial connections.
Per the image above, you have a whole host of integrins expression in white blood cells, your immune cells, and these will be fairly important soon. They also have an extensive role in many forms of pathology, from cancer to heart disease, many forms of fibrosis, and finally auto-immunity, these are all covered in the paper above, and even if you are not a science person, I advise you to glance over those sections. The following image is an example of the whole of integrins and cancer. Integrins can both start and accelerate tumor growth (metastatic cascade).
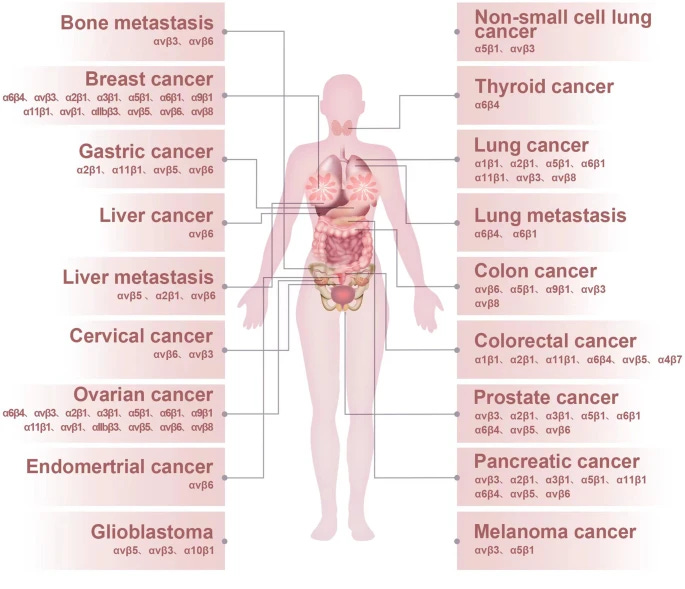
If this short but rather complicated introduction is out of the way, here is the significance of Integrins in regard to SARS-CoV-2.
Biological and Clinical Consequences of Integrin Binding via a Rogue RGD Motif in the SARS CoV-2 Spike Protein
Even as early as 2021, the role of Integrins as possible secondary mediators of SARS-CoV-2 infection (bypassing the use of ACE2) was proposed, and so was argued all the potential effects that this possible interaction might set-off, off because as many of my readers are aware by now, the interaction of SARS-CoV-2 with “unusual” receptors modulate their presence, sometimes making you produce much more of it, sometimes a lot less, and both have clinical significance, this is a recurring theme with this virus, which basically takes advantage of your of the loss of equilibrium to continue its cycle, what I called “Homeostasis weaponization” also in 2021.
As I wrote in other substacks, many variables come into play in regards to protein interactions, not only the sequence (the amino acid letters) but also the shape and position of the protein, so simplify it. In this particular case, the region that interacts with integrins is the RGD one, and the authors of the paper compared this RGD motif to thousands of other protein sequences and found that the vast majority don’t participate in this integrin-mediated signaling, most are “locked” in a position that effectively makes it impossible, but that is not the case with SARS-CoV-2, while its RGD motif is not completely exposed, the position it is in is flexible enough to allow the interaction.
The paper goes on to bring to light the potential interactions of specific integrins with platelets, the potential for coagulatory problems, and also angiogenesis, the last is a particular interest not solely based on the potential to initiate cascades but angiogenesis among other pathologies is correlated with cancer (a point I am bringing up, not the authors).
Evidence later in 2022 showed us that the Spike, the novel rogue RGD motif in the Receptor-Binding Domain is a functional integrin agonist, meaning not only it interacts with integrins, it makes your body produce more of it, specifically αvβ3 and αvβ6.
On my birthday last year, a paper was published, one that caught my eye instantly.
SARS-CoV-2 Spike triggers barrier dysfunction and vascular leak via integrins and TGF-β signaling
Here, we investigated whether SARS-CoV-2 S contributes to endothelial and epithelial barrier dysfunction in vitro and vascular leak in vivo. Our study reveals that full-length S and the RBD from SARS-CoV-2 are sufficient to mediate barrier dysfunction and vascular leak in an ACE2-independent manner. Further, transcriptional analyses showed that S modulates expression of transcripts involved in regulation of the extracellular matrix (ECM), and experimental validation revealed a mechanism in which glycosaminoglycans (GAGs), integrins, and transforming growth factor beta (TGF-β) signaling are required for barrier dysfunction. Finally, we find that SARS-CoV-2 infection in vivo triggers vascular leak in the lungs of mice, which is reversed by antagonizing integrins. These data uncover the role of SARS-CoV-2 S in promoting barrier dysfunction and provide critical mechanistic insight into this process.
Since we are barely at the start of the reason I decided to write this substack, I will just share the highlights of this paper and attempt to simplify it, although a lot of attention should be brought on to the mechanism described in the quote above, in relation to TGF-β, quite a few of the integrins that play a major role in regards to this quite potent protein are effectively modulated by the Spike protein, and this is the de facto “Theo’s pathway”, it is a unique pathway that follows a different molecular cascade, but ends up with similar outcomes that I have covered in my Substack publications.
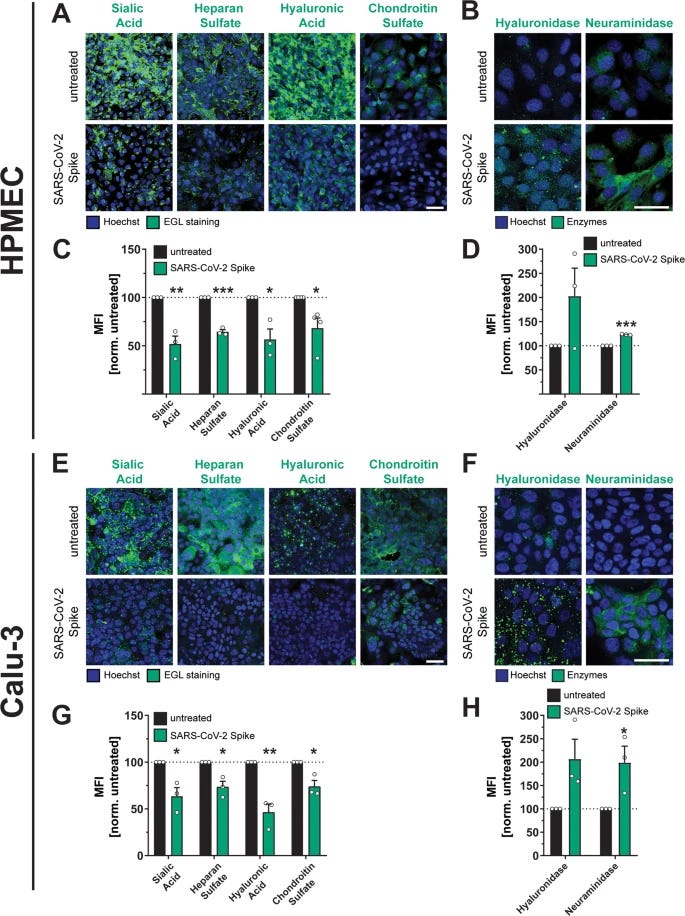
The authors test their primary observations and attempt to elucidate if and how the SARS-CoV-2 Spike protein (S) mediates endothelial and epithelial hyperpermeability, changes in the permeability of anything, from cells to tissues can initiate entire cascades that lead to different types of damage, from inflammatory responses to actual damage to organs. With intricate testing, authors found that SARS-CoV-2 S and RBD (Receptor Binding Domain) can facilitate the aforementioned state, and they even compared with other two CoV (HCoV-229E and HCoV-OC43), and this effect is particular to SARS-CoV-2, only.
Authors also found that the S triggers the disruption of the endothelial and epithelial glycocalyx layer, a protective layer made of small sugar molecules (simplified) both free S and virion-associated S could trigger this response, important because you get free S from mRNA by the “tons”.
To further develop their hypothesis, they tested if the Spike could mediate vascular leak in vivo, and by administrating Spike intranasally they found that S significantly induce vascular leak in the lungs as well as spleen, small intestines, and to a minor level liver and brain. This is in line with almost the entire body of scientific literature and clinical evidence so far in regards to a sizable portion of the SARS-CoV-2 infection dynamic, where severity is what dictates the level of damage and tropism (which organ the virus is able to “infect” and damage), with brain and liver often being the last ones, excluded outliers. And finally finding that these mechanisms are mediated by integrins.
And now, to the main (kinda) reason I chose to write this now.
Lymphocyte integrins mediate entry and dysregulation of T cells by SARS-CoV-2
Some evidence suggests the infection of lymphocytes by SARS-CoV-2, including colocalization of SARS-CoV-2 spike (S) protein and lymphocytes in lung tissues of COVID-19 patients and strands of SARS-CoV-2 sub-genomes in immune cells from bronchoalveolar lavage fluid (BALF) and sputum samples of severe COVID-19 patients.2 Angiotensin-converting enzyme 2 (ACE2) and some other reported S protein receptors are barely expressed in lymphocytes. Some recent studies suggest that integrins may act as SARS-CoV-2 receptors, shedding light on the dysregulation of lymphocyte functions by SARS-CoV-2. However, a clear-cut mechanism underlying the crosstalk between SARS-CoV-2 and lymphocyte integrins remains elusive.
So after quite a while we now effectively know the mechanism by which SARS-CoV-2 can infect T Cells
Months ago a paper made the rounds everywhere, a paper that was severely mischaracterized by many (incidentally what made me start blocking a lot of other people) in which the authors found that SARS-CoV-2 could infect T Cells-Lymphocytes.
While the authors lacked a proper, specific entry mechanism, their overall idea and observations were not merely correct, they are pertinent to this day. Taken together this explains a lot of the immune dysregulation we see in regard to the T Cells. And some of their proposed mechanisms are highly pertinent here. Remember that I said integrins are complex and contextual ? All the following are examples of how complex integrins become.
In the Substack above, the proposed mechanism for the death of T Cells are ROS-Hypoxia-related pathways. Among other integrins, Hypoxia-inducible Factor Regulates αvβ3 Integrin Cell Surface Expression, Hypoxia can regulate our old friend Galectins, especially one, but recently they found it can also regulate Galectin-3.
Integrin αvβ3 is considered one of the biggest Galectin-3-binding proteins, but more importantly, Galectin-3 when found outside the cell or exogenously added can bind to integrins and facilitate cell trafficking and activation. Galectin-3 can also bind to αvβ1, αvβ5 and αvβ6 in a glycosylation-dependent manner, meaning depending on how sugar molecules are added, Gal-3 can interact with these integrins.
In the end, one of the most overlooked, under-researched aspects of SARS-CoV-2 is its N-Terminal Domain, with its known region called Galectin-fold which is a very close mimic of human Galectin-3, a protein highly expressed at many different levels of the infection, and given its chimeric nature, and by what we covered in this substack, arguably one of the most impactful. And to connect another one of my “favorite” topics.
Integrins play an important role in both endotoxin states and sepsis, and here one of the most mentioned integrins is responsible for the chronic inflammatory processes in many conditions and one that could explain why and how the Spike is an “integrin” agonist. The Endotoxin interaction with the Spike is arguably the most important aspect of the severity of disease and bad long-term outcomes, now becoming more evident as we find more and more evidence.
Now, the good news.
The D405N Mutation in the Spike Protein of SARS-CoV-2 Omicron BA.5 Inhibits Spike/Integrins Interaction and Viral Infection of Human Lung Microvascular Endothelial Cells
Here we show that the D405N mutation is mainly expressed in the Spike of all the recently appeared SARS-CoV-2 Omicron subvariants. From a phylogenetic analysis of SARS-CoV-2 variants, we identified in April 2021 an Omicron sublineage, Omicron BA.2, in which a D405N mutation is dominant. The switch from RGD to RGN induced a dramatic loss of function by completely inhibiting SARS-CoV-2 infection of ACE2-negative HL-mECs. This evolutionary trajectory was imposed on the newly emerged Omicron subvariants by the hostile microenvironment generated in response to Omicron BA.1 infection [24] and aimed at preserving viral fitness. Therefore, as unexpectedly appeared in the first SARS-CoV-2 pandemic virus, the RGD motif and its integrin-binding activity disappeared during virus evolution, along with the SARS-CoV-2 capability to infect HL-mECs and to promote endothelial dysfunction. This finding highlights this initial constraint as not being more necessary for further virus adaptation to the human host and, at the same time, supports the early key role of integrins as the main receptors for SARS-CoV-2 entry in ACE2-negative cells. The role exerted by integrins in ECs as alternate receptors to ACE2 for SARS-CoV-2 has been further confirmed by others
A single mutation at 405 changed the RGD motif to RGN inducing a substantial loss of function, and basically almost completely stopping the pathological effects of integrins in regards to SARS-CoV-2 and the endothelial dysfunction discussed above. Authors argue this is one of the main reasons the “new” variants are rather mild, in this case, BA.5.
My personal opinion, echoing the same observations from other papers I covered months ago is similar, while severely diminished, there should be research into what the new variants are doing to immune cells, even while in the presence of severe loss of function, there could still be other mechanisms by which the virus could cause immune dysregulation, especially long-term. And especially among the vaccinated.
As a final note, I have covered many of the mechanisms mentioned here, such as hypoxia, endotoxin, ROS, and mitochondrial dysfunction, I just didn’t want to link most of them here for length's sake.
As always, I appreciate the subscribers who choose to support this substack in any way they see fit.







Thanks John, another awesome write up and one that was in my to-do list.
The angiogenesis - cancer link is worthy of follow up.
More on effects on the glycocalyx, which can take months or years to recover:
"Persistent capillary rarefication in long COVID syndrome"
https://doorlesscarp953.substack.com/p/persistent-capillary-rarefication
I assume this means that free floating spike can bind to t-cells via these integrin receptors, correct? What would happen to the t-cells since there isn't a virus to infect them?