The theme song for this one, warning it is atmospheric black metal, on the heavy side, the first song especially beautiful. The cover is an actual photograph from WWI.
My intention was to finish my new Beyond Mathematical Odds and publish it today, but this paper just published in Cell is just too important given the overall dynamics at play to pass. I wrote many months ago about a specific paper that I found particularly important long-term.
It led me to write this one after, even more important because you can substitute SARS-CoV-2 for any infection, acute, chronic, or long-term low-grade inflammation, a state untold millions find themselves in.
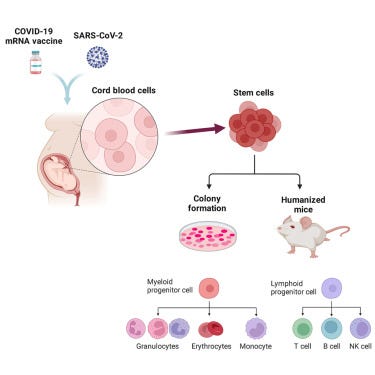
Skewed Fate and Hematopoiesis of CD34+ HSPCs in Umbilical Cord Blood Amid the COVID-19 Pandemic
SARS-CoV-2 infection/vaccination correlate to the in vivo fate of CD34+ HSPCs.
SARS-CoV-2 infection/vaccination correlate to hematopoiesis of CD34+ HSPCs.
SARS-CoV-2 vaccination correlates with CD34+ HSPC numbers/frequencies in the UCB.
Per the highlights this is in relation to both infection and vaccination, per many of the other pieces I covered/wrote, which is good, so there isn’t the intense or one-sidedness.
Umbilical cord blood (UCB) is the blood originating from the neonate. UCB differs from that of adult peripheral blood, in which UCB contains higher numbers of monocytes and nucleated red blood cells (RBCs), and lower numbers of matured RBCs and T cells, especially CD8+ T cells . Lymphocytes in UCB produce fewer absolute levels of cytokines and have higher abundance of anti-inflammatory cytokines than adult peripheral blood sources. More importantly, UCB is highly enriched with multipotent hematopoietic stem progenitor cells (HSPCs) as identified by the surface expression of CD34 molecules, which are essential for the maintenance of the bone marrow and blood systems; HSPCs have been widely used for various therapeutic and research purposes, such as bone marrow transplants and the treatment of severe cases of COVID-19. Whereas the fate and functionality of CD34+ HSPCs are severely affected by the host’s health status, such as infection by a pathogen, and can cause stress-induced hematopoiesis in these HSPCs.
For clarification, CD34 is a transmembrane glycoprotein and a marker to measure different biological effects, a transmembrane “anything” means it is distributed in the entire membrane and it is like a small send/receive gate in the cell. Per what is described here these cells are important not only for anything Stem cell-based but out of the scope of this paper, for the overall immune health of any person (more on this later in this piece).
For clarification I will need to interject a lot on this one, so bear with me.
Infection of CD34+ HSPCs by SARS-CoV-2 in vitro displayed reduced hematopoietic colony formation potential and an increased potential for erythrocyte and thrombocyte precursors as well as higher susceptibility to apoptosis.
Striking changes were observed in the CD34+ cell fraction ― the total numbers of CD34+ cells drastically reduced 4-fold in the vaccinated donor group, in which this change was correlated to the induction of apoptosis in CD34+ cells, likely mediated by IFN-γ-related pathways as determined by total transcriptome assay. In addition, the hematopoietic abilities of these CD34+ cells showed skewing in two different hematopoiesis assays – an in vitro colonyformation unit (CFU) and a mouse humanization assays - as represented by high T cell/B cell ratios and higher erythrocyte & lower granulocyte-macrophage colony formations.
These data indicate that both previous SARS-CoV-2 infection and/or vaccination impair CD34+ HSPCs quantitatively and qualitatively by stress-induced hematopoiesis, which is a great concern in the collection as well as the utilization of UCB as a source of CD34+ HSPCs used in/for future therapies, treatments, and research.
Results:
Both previous-SARS-CoV-2 infection and vaccination negatively impact CD34+ HSPC frequencies and numbers in the UCB.
No significant difference in the baby’s weight at term as well as the volume of UCB were observed between groups, but the numbers of mononucleated cells (MNC) decreased in the non-vaccinated and vaccinated donor groups, even so more in combination with the vaccination and the presence of anti-N IgG more than 2-fold compared to the negative donor group.
A striking difference was observed in both the % frequency and the total estimated number of CD34+ cells per 1 mL of UCB ― both parameters decreased 1.7 to 3-fold in the non-vaccinated and vaccinated donor groups, respectively, compared to the negative donor group, regardless of the positivity for IgGs against SARS-CoV-2 S and N proteins
Using different groups and different ways to measure the time of infection, here they found what anyone who reads scientific literature in regards to SARS-CoV-2 is aware of, both vaccination and infection bear a cost, but I find soothing for a few seconds that they bring evidence to one of my points, ages old point by now. Breakthrough infections carry a more significant decline than either infection or just vaccination, twice the decrease compared to the ones never infected.
To assess the recovery time needed for decreased CD34+ cell numbers of donors following the vaccination/infection to baseline as established by the negative donor group, the % frequencies and numbers of CD34+ cells per 1 mL of UCB against the days post and vaccination were plotted. Both parameters were inversely correlated to the term following the vaccination, though there was no significance difference by the positivity for IgGs against SARS-CoV-2 S and N proteins, indicating that these impacts on CD34+ cells were largely caused by the vaccination, in which the trends continued without recovering to baseline over the entire gestation period.
Read this section as much as needed until it dawns upon you. The effects of decreasing CD34+ cell numbers were more pronounced among vaccinated rather than by infection status, meaning the stark differences in the reduction effect are caused by the vaccination. This is a solid argument for the HSCs paper I covered months ago, in which they demonstrate the negative effect the Spike Protein has on Stem Cells, and bring to the attention that perhaps the vaccines using Spike Protein could damage these cells too.
Following this the authors explain they did not find detectable levels of both N antigens or free S proteins, indicating that these changes were not derived from viral S of vaccine Spike, but these cells were becoming apoptotic (dying). Furthermore, there were no differences in cytokines in the plasma from Umbilical Cord, but one of their tests suggested the involvement of Interferon-Gamma, an old acquaintance around here.
Taken together, these data suggest that the IFN-γ-related signaling pathways promoted by SARS-CoV-2 infection could involve in the induction of apoptosis observed in CD34+ HSPCs and thus might affect the fate and survivability of CD34+ HSPC populations in the UCB.
A point of pride very early on, in the mRNA vaccine implementation was its (skewed) immune response and its broad, potent Interferon-Gamma response, a remarkable response “insert sales pitch here”. The pathway argued by the authors, which has precedence and evidence in literature is even more present among the vaccinated mothers, more pronounced after the second dose of the vaccine, and it lasted the entire gestation period.
While out of the scope of this paper, and not argued by the authors, this goes merely beyond creating a shortage of HSPCs for research, treatment, and possible new onset of hematological issues in HSP cells recipients, this clearly has the signs of premature aging of the immune system, the so-called “inflammaging”. A hallmark of this state in old people is the skewed immune system, and among many other aspects, a higher level and presence of Interferon-Gamma.
In the scope of the study alone, this already put to rest the “vaccination in gestating mothers is protective/harmless”, but I will go beyond it. From here.
Collectively, these data suggest that both IFN-α and IFN-γ directly stimulate HSC proliferation, and, if the exposure is prolonged, result in a loss of repopulating activity. IFN signaling also appears to play a negative role in regulating HSC quiescence and repopulating activity under basal conditions. Of note, there is evidence (at least for IFN-γ) that IFN signaling regulates HSC function in humans. Specifically, Yang and colleagues showed that treatment of CD34+ CD38− human cord blood cells with IFN-γ markedly inhibits their ability to support multi-lineage hematopoiesis when transplanted into NOD-SCID mice
I could keep citing literature, but for simplicity and time's sake I will finish the Gamma section here, the effects of chronic infection are extensive, and chronic stimulation of Interferon Gamma leads to a whole host of immunological changes, I would like to remember that this Interferon also modulates the presence of IDO, the enzymes that break down Tryptophan, and is a major player in the Kynurenine Pathway. Interferon Gamma also played a central role in a recent paper talking about Thymic atrophy in regard to SARS-CoV-2 infection.
And since the SARS-CoV-2 Spike as an Endotoxin Delivery me and two other friends have looked extensively down this route, because it is as clear as day that this play a major role in almost every single facet of this whole mess, either by canonical or non-canonical pathways. If the Spike Protein is Frankenstein, the capacity to modulate Endotoxins and biofilms are the body parts that constitute the monster.
Chronic exposure to a TLR ligand injures hematopoietic stem cells
Hematopoietic stem cells (HSC) can be harmed by disease, chemotherapy, radiation, and normal aging. We show in this study that damage also occurs in mice repeatedly treated with very low doses of LPS. Overall health of the animals was good, and there were relatively minor changes in marrow hematopoietic progenitors. However, HSC were unable to maintain quiescence, and transplantation revealed them to be myeloid skewed
Early severe impairment of hematopoietic stem and progenitor cells from the bone marrow caused by CLP sepsis and endotoxemia in a humanized mice model
Given everything I have written in this Substack, it is objectively true, and factual that the viral infection to a lower degree, the vaccination to an unparalleled degree has brought drastic changes to a third of the global population, and while I regard myself a competent complexity “guy”, we just can’t fathom the immunological shifts and evolutionary changes on pathogens this will set off.
We are in uncharted territory, and have been for a while now.
Before ending this piece, already depressing enough given what it entails on its own, I want to bring the following paper to my readers attention.
Impact of SARS-CoV-2 on the microbiota of pregnant women and their infants
The microbiome inherited at birth exerts marked effects on immune programming with long-term health consequences. Here, we demonstrated that the gut, vaginal, and oral microbial diversity of pregnant women with SARS-CoV-2 infection is reduced, and women with early infections exhibit a different vaginal microbiota composition compared to healthy controls at the time of delivery. Accordingly, infants born to pregnant women with early SARS-CoV-2 infection exhibit a unique oral microbiota dominated by Streptococcus species. Together, we demonstrated that SARS-CoV-2 infections during pregnancy, particularly early infections, are associated with lasting changes in the microbiome of pregnant women compromising the initial microbial seed of their infant. Our results highlight the importance of further exploring the impact of SARS-CoV-2 on the infant’s microbiome-dependent immune programming.
Changes in the microbiome in early life can dictate not only immune responses, but even behavioral changes until early adulthood, and the same applies in infants, this is solely based on your microbiomic changes. I intend to cover this in the near future, but for the curious or literate in microbiome, this is a good starting point. The following excerpt should give you a hint “unique oral microbiota dominated by Streptococcus species”.
And once again, what the virus does, the Spike does it faster. And to put to bed the synthetic antibody salesman pitch, the following.
I wish you all a nice Sunday. I plan on writing on things outside subjects covered so far soon, and personal experiences, given where things are headed as a whole.
I welcome and appreciate the support of those who choose a paid subscription, or who decide to buy me a coffee whenever they feel like it, and everyone who shares my Substack. Without all of you, this wouldn’t be possible.







Red pill alert needed. I think I need a drink, I feel as I did after reviewing the myriad ways to initiate autoimmune disorders and the consequences. Crashed CD34 out to 300 days with no uptick. RSV in kids you say? No sh!t.
Pure evil, thanks John. I've shared of course with Genervter.
Measles appears milder long term, but the consequences of CD34+ suppression are of interest here:
Targeting and Hematopoietic Suppression of Human CD34+ Cells by Measles Virus†
...Herein we present evidence that measles virus (MV) disrupts hematopoiesis by infecting human CD34+ cells and human bone marrow stroma. MV infection does not affect the hematopoietic capability of hematopoietic stem cells (HSCs) directly; rather, the infection impairs the ability of stroma to support development of HSCs. These results suggest that MV-mediated defects in hematopoiesis contribute to the long-term immunosuppression seen in measles.
..One hallmark of MV immunosuppression is profound lymphopenia (including that involving CD4+ T cells, CD8+ T cells, and B cells), monocytopenia, and neutropenia that persists following virus clearance (2, 20, 33). Interestingly, young children with the highest level of BM output (24, 28) recover most quickly from MV-induced lymphopenia and immunosuppression. Older children and adults with a less robust BM reconstitution capacity require longer periods of time for recovery from MV immunosuppression.
...Other immunosuppressive viruses, such as human cytomegalovirus, dengue virus, murine leukemia virus and human immunodeficiency virus type 1, have been reported to target BM stroma and inhibit hematopoiesis by reducing expression of supportive cytokines such as stem cell factor and/or increasing expression of suppressive cytokines such as transforming growth factor β (1, 4, 21, 22, 27, 30, 41). Thus, stromal cell targeting may be a common mechanism used by immunosuppressive viruses to induce hematopoietic suppression.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC136250/