Sequential infection, inflammaging and the microbiome
And how taurine helps it
If you want to skip the already simplified science, just use the Control/Command+F (or find a feature in mobile browsers) and search for “Why is this important”.
And since this became longer than planned, Taurine is one of the most promising “treatments” to what is discussed here, you can find more in the Additional Resources section or here.
Before going any further on the other subjects I have been writing on and planning to publish, such as HERVs and misfolded proteins, it is more beneficial to build further context, after all, I see this publication as one big piece of the puzzle and while you can read most articles as standalone, it is more beneficial to “connect the dots”. And within recent such as the impact of vaccines on the microbiome, and now Inflammaging. I will summarize both the concept of Inflammaging and the referred article below.
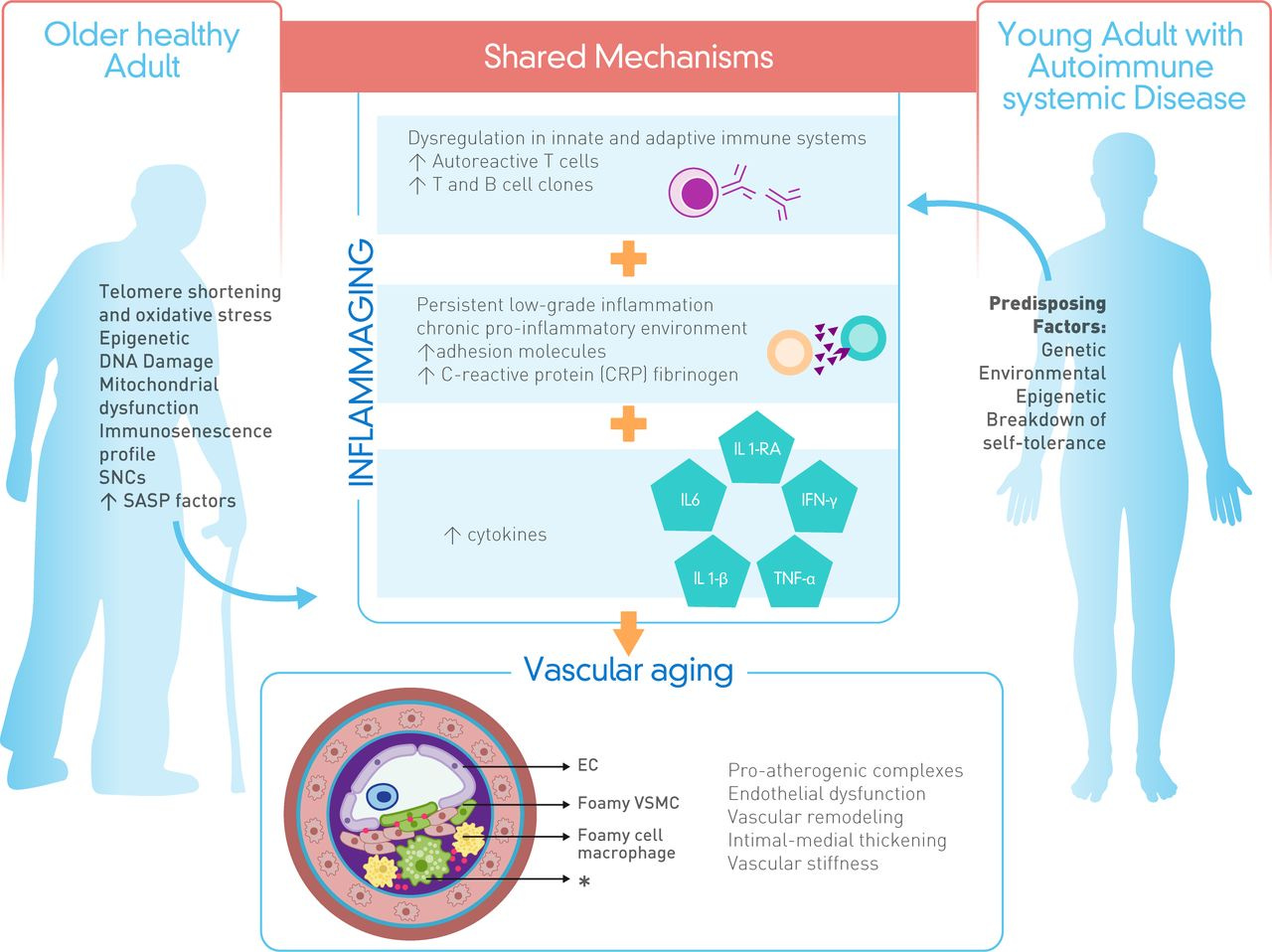
Inflammaging is often associated with older age and the physiological state the body finds itself in, with persistent low-grade inflammation, and a skewed and slower metabolism, contributing significantly to poorer immune function. At a minimal level, this is a “natural” state of aging, but the modern lifestyle contributes disproportionally to transforming a minor, “normal” state into a societal-level problem.
The concept of inflammaging is a recent one, and given its recency a lot of interest and research have been put into studying it. We now understand this isn’t merely an “old age” problem, or chronic disease-related problem, but a problem that happens over time, the accelerated “aging” of our immune system. And the microbiome plays a very significant role here.
My substack article referred to above talks about how even small amounts of inflammation, in the paper, found that with each additional round of “inflammation,” a decrease in your hematopoietic stem cell production, regeneration, and differentiation, even after a long recovery time. Repetitive induction of inflammation leads to a progressive depletion of functional HSCs with little regeneration during recovery periods.
Meaning exposure to inflammation even in early life affects your immune system later in life. Each part of this complex puzzle plays a part, the gut microbiome (dysbiosis), low-grade inflammation, new infections, and failure of bioenergetic pathways all create feedback loops leading to these problems. This recent substack is a nice addition to what we are about to both discuss and evidence to what I call “short-term gains, long-term pains” of vaccination.
Sequential early-life viral infections modulate the microbiota and adaptive 2 immune responses to systemic and mucosal vaccination
This quite timely paper focuses mostly on developing and discovering to find a reliable model of sequential infection for laboratory use, as well as the microbial and immune changes from these infections. They decided to use purely a virus model, which from my perspective is a better model to understand such changes, given that adding bacterial or fungal infections adds so much complexity and confounders (one thing adds to another, that adds to another, and you don’t know which is which) this is a better way. The model in question.
We sought to develop an “immunologically-mature” murine model in a genetically 109 defined background by utilizing a well-controlled series of microbial exposures, focused 110 exclusively on viruses, that could be rapidly administered to permit further experimental 111 intervention by the age of 10 weeks. Thus, we inoculated C57BL6/J mice with six distinct viruses - murine rotavirus strain EDIM-Cambridge (MRV, 104 112 ID50 by oral gavage), murine gamma-herpesvirus 68 (MHV68, 105 113 PFU intranasally), murine norovirus strain CR6 (MNV, 106 114 PFU orally), influenza virus strain PR8 (IAV, 300 PFU intranasally), murine astrovirus (MAstV, infectious fecal filtrate by oral gavage), and coxsackievirus B3 (CVB3, 108 115 PFU orally) 116 - at 1-week intervals beginning when pups were 7 days old
As one would expect after suffering from multiple sequential infections, the authors found the levels of certain pro-inflammatory proteins were higher in the mice after 9 weeks of exposure, these proteins being Interleukin-6, Interleukin-12p70, Interferon-Gamma, and Tumor Necrosis Factor Alpha. A normal physiological response to infection with any pathogen.

Their focus was to study the changes in sequentially infected mice, so their choice of biomarkers to measure these changes is related to immune function, it is not as comprehensive as I would like, and it does not have markers to measure inflammaging or was even the goals. We still can gather useful information for this discussion, they found a significantly higher presence of LinSca1+c-Kit+ hematopoietic cells, which become other cells.
Incidentally, they found that certain B cells, such as immature B cells, B1 B-Cells, and plasma cells were present at lower levels, and Germinal Center ones at higher levels. Also, they found a loss of T Regulatory cells. It would be really beneficial for the understanding of multiple infections and how/when one starts shifting towards inflammaging, the accelerated aging of the immune system. The information presented so far is enough to deduce this effect.
Loss of B1 cells, and a tilt towards a lower presence of immune regulatory cells, are both a hallmark of an aging immune system and aging of the cells, but also a sign of autoimmunity, the change in immune cells also indicates a higher antibody affinity. Another aspect of inflammaging.
If you don’t recall B1a B Cells also are a very important part of the immune axis responsible for defending your body against fungal infections.
But the reason I decided to write this article as the following section, is highly important in the long-term perspective.
Sequential viral infection modulates the intestinal microbiome
To further explore potential differences in the bacterial communities between mock and sequentially-infected groups, we performed Kruskal-Wallis rank sum tests and linear discriminant analysis (LDA) based on ASV abundances. These analyses revealed differentially-abundant taxa between the two groups). Specifically, the relative 290 abundances of Duncaniella (p = 0.001, fdr = 0.023), Clostridium sensu stricto (p = 0.011, fdr = 0.007), and an unclassified genus belonging to the Barnesiellaceae family (p = 0.0007, fdr = 0.019) were higher in the intestinal microbiota of sequentially-infected mice ). Conversely, the intestinal microbiota of sequentially-infected mice exhibited reduced relative abundances of Paramuribaculum (p = 0.007, fdr = 0.058), Turicibacter (p = 0.002, fdr = 0.031), Alistipes (p = 0.007, fdr = 0.058), Oscillibacter (p = 0.048, fdr = 0.197), Schaedlerella (p = 0.035, fdr = 0.153), Kineothrix (p = 0.024, fdr = 0.119), Bifidobacterium (p = 0.0004, fdr = 0.018), and an unclassified genus belonging to the Prevotellaceae family (p = 0.023, fdr = 0.119) (Figure 5D and S6C). In the CNT, only the relative abundance of Faecalibaculum (p = 0.012, fdr = 0.47) was decreased in sequentially-infected mice
Mice that were sequentially infected experienced a change in their microbiome, and this change further modulated the immune response against vaccination, especially within the specific route, when exposed to vaccination via intramuscular (IM) they had diminished antibody response, and as expected no IgA, antibodies that are the hallmark of mucosal immunity. When vaccinated intranasally (IN) the mice had IgA but the sequentially infected ones also had a diminished response.
Overall, these findings indicate that repeated viral exposures impact responses to vaccination by blunting vaccine-specific antibody responses and potentially enhancing antigen-specific T cell responses particularly following mucosal exposure. These findings highlight the importance of considering exposure history in evaluating pre-clinical interventions, as well as the immunological consequences of early-life viral infections.
This paper gives us a lot of information, and a lot to think about, it would be incredibly beneficial if a follow-up study was done following longer-term consequences of these immune responses, especially the effect on the thymus. (I said it once, and repeat, it is pretty easy for me to say something while not running the experiments, designing them, writing the paper, so anything here isn’t a criticism).
Consequentially extrapolating from the information presented, based on the data, we can deduce that there are many degrees of accelerated aging of the immune system going on, and if done in humans or human models, longer term we would observe a higher incidence of autoimmunity, among immune loss (weaker immune system), the persistent high level of Interferon Gamma itself alludes to that. Good to have a short-term, double-edged sword that immune suppresses you medium-term.
Why is this important ?
The degree of complexity between the microbiome, our metabolism, immune system can’t be stated enough, so understanding how vaccines, and numerous infections throughout life impact the immune system, the microbiome, inflammatory levels is of extreme importance by one simple fact.
This is what happening at a global level, with obviously greater incidence among the people vaccinated for Covid. It is a vast sea of contributing (confounding) factors, a complex equation with many variables, but the result of this complex math is the accelerated aging of the immune system. Most of my frequent readers if not the majority know someone who battles an infection almost every other month, and the list of problems goes on.
Consequential infections from different classes of pathogens possess other long-term problems, my focus for most of the year has been Endotoxin and its response, thus leading to Endotoxic Tolerance, but that is merely just the most immune-tolerant state studied, in fact, one can say there is a receptor tolerant state to ALL other receptors, Endotoxin Tolerance is TLR4 tolerance. And the other receptors, when found in a tolerant state have rather… unpleasant long-term effects.
Additional resources
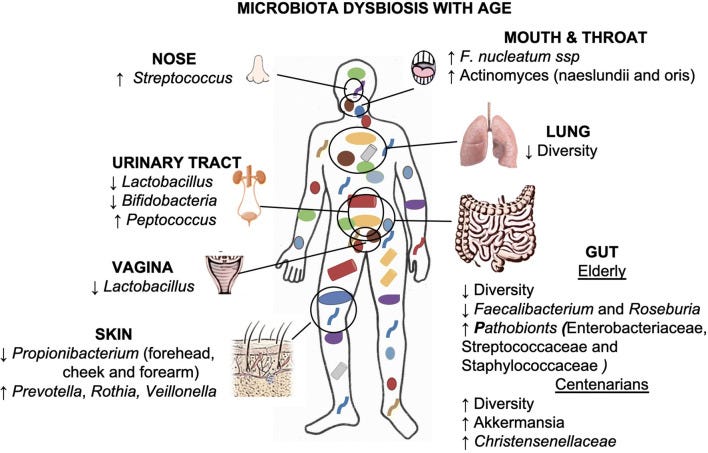
A hallmark of getting old, especially in the modern world is the loss of immunocompetence (poor diet, low nutrition, almost no exercise, stress, pollution, obesity), leading to many of the aspects discussed here. Given the role of the microbiome, aging is also associated with a change in the microbiome composition, and it can either help to avoid or help to induce damage, so it plays a role in inflammaging.
Dysbiosis (the loss of microbiotic equilibrium) plays a central role here.
Cytokines including TNF-α, TGF-β1, IFN-γ, IL-1β, IL-6, IL-17A, IL-10, and VEGFA were detected…
The results showed that obesity influenced metabolic variables (including fast/fed glucose, insulin, and triglyceride), retinopathy and nephropathy, and the gut microbiota. Obesity mainly reduced the ratio of Bacteroidetes/Firmicutes and influenced relative abundance of Proteobacteria, Actinobacteria, and Spirochetes. Obesity also increased intestinal permeability, metabolic endotoxemia, cytokines, and VEGFA.
Endotoxins are known to be one of the greatest contributors to inflammaging, among almost every other modern disease. Lysosomes are the “recyclers” inside cells, and failure in lysosomal function is not only related to aging, but to a myriad of different diseases, both chronic, inflammatory, or autoimmune.
Chronic systemic inflammation is one of the hallmarks of the aging immune system. Here we show that activated T cells from older adults contribute to inflammaging by releasing mitochondrial DNA (mtDNA) into their environment due to an increased expression of the cytokine-inducible SH2-containing protein (CISH). CISH targets ATP6V1A, an essential component of the proton pump V-ATPase, for proteasomal degradation, thereby impairing lysosomal function. Impaired lysosomal activity caused intracellular accumulation of multivesicular bodies and amphisomes and the export of their cargos, including mtDNA. CISH silencing in T cells from older adults restored lysosomal activity and prevented amphisomal release. In antigen-specific responses in vivo, CISH-deficient CD4+ T cells released less mtDNA and induced fewer inflammatory cytokines. Attenuating CISH expression may present a promising strategy to reduce inflammation in an immune response of older individuals.
Another aspect of inflammaging is the abundant expression of receptors related to immune responses, such as Toll-Like Receptor 4 (the one responsible for sensing endotoxins/LPS) among the other receptors of the family, thus leading to a vicious cycle of inflammation. A proposed, and successful intervention in the referred paper (time and tested for decades, one could say thousands of years even) is Dietary Restriction, meaning consuming fewer calories but with proper nutrition. This is different from Fasting.
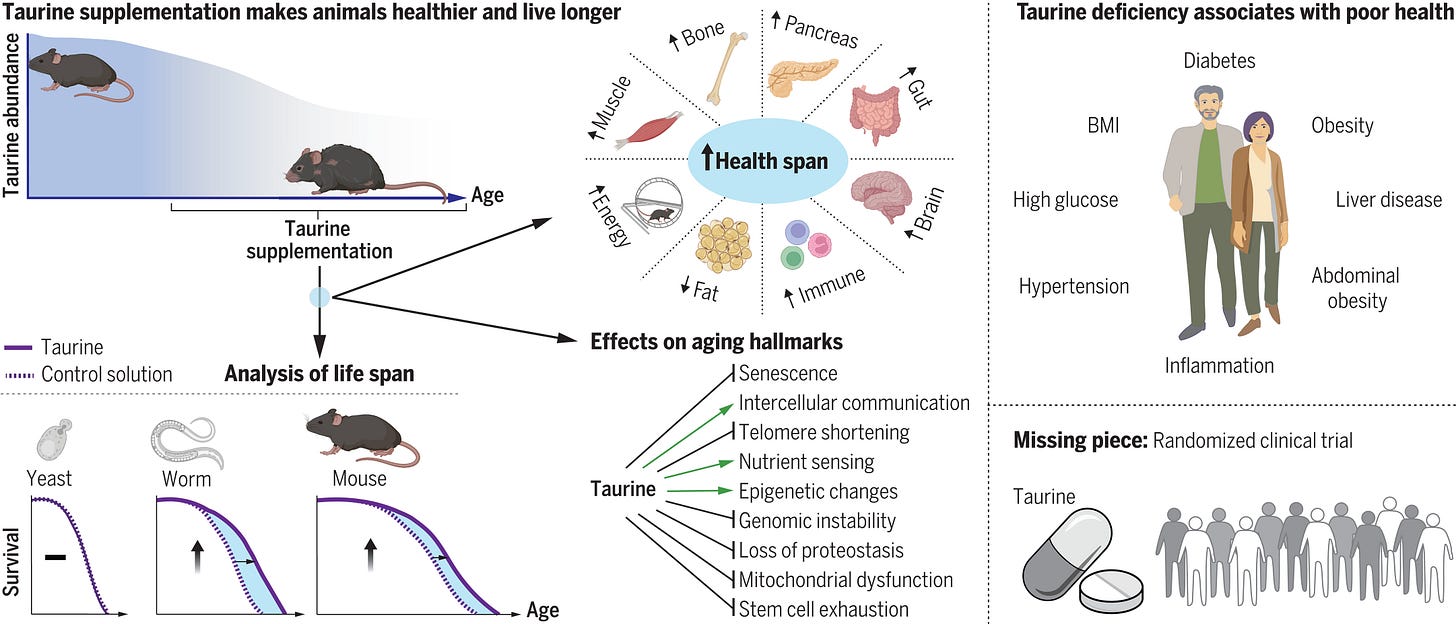
A recent and promising intervention for aging and its effects on the immune system (thus all the cascade that implies) is Taurine.
Taurine reduced cellular senescence, protected against telomerase deficiency, suppressed mitochondrial dysfunction, decreased DNA damage, and attenuated inflammation. An association analysis of metabolite clinical risk factors in humans showed that lower taurine, hypotaurine, and N-acetyltaurine concentrations were associated with adverse health, such as increased abdominal obesity, hypertension, inflammation, and prevalence of type 2 diabetes. Moreover, we found that a bout of exercise increased the concentrations of taurine metabolites in blood, which might partially underlie the antiaging effects of exercise.
CONCLUSION
Taurine abundance decreases during aging. A reversal of this decline through taurine supplementation increases health span and life span in mice and worms and health span in monkeys. This identifies taurine deficiency as a driver of aging in these species. To test whether taurine deficiency is a driver of aging in humans as well, long-term, well-controlled taurine supplementation trials that measure health span and life span as outcomes are required.
Taurine supplementation in older adults helps recover muscle, lowers inflammation, and helps with oxidative stress, it also directly helps older people get more cognitive and physical benefits from exercising. It also possesses benefits for cardiovascular health.
For the normal person, the best approach is just fixing your diet, the best taurine sources are animal protein, dairy, and seaweed too. People with poorer diets, or specific issues such as gut inflammation can benefit from the supplement itself. If you recall my recent article on exercise, you will remember I said the problem with older people is that their bodies lose the ability to properly digest animal protein, so besides fixing their diet, they should definitely add the supplement.
In my Long Covid biomarkers article, you will find that Taurine is also significantly diminished in those patients. Although I personally hold the opinion that most people afflicted with Long Covid will benefit more from Creatine, and get Taurine from their diets unless they fit into the ‘specific issues’ part. I rather not have people on 10+ supplements per day unless strictly necessary.
The next one, a very short one, is an update on matters that are important and of interest, both to long-term health and to myself. After that…well…you will see.
PS: If money isn’t exactly a problem, peptides can basically fix most of these issues, why I am mentioning them here, and now ? Because the FDA, after months, is clamping down on the market hard, it may or may not affect the overall supply, but definitely will affect the price
If you chose to support me, or supported me at any point, thank you. And thank you for reading.




The point here is in the text, but to simplify further. The last 3 years created a cascade of events that is accelerating the aging of the immune system of the majority of the world's population, with the mRNA injected a lot further down the road.
Everything discussed here is directly correlated with the expression of the bad HERVs, and soon you will understanding why that is important. What is the other hallmark of aging ? Misfolded proteins.
Any sense of dosing of supplement to start with?