Microclots and Protein modification in Long Covid and POTS
Biochemical Cascade
Last hour addedum.
Since my last article, written on the impending supply chain shock and growing shortages that may come our way, I have paid extremely close attention to the Iran War, specifically from that perspective. I didn’t have in me to write anything else, as the situation evolved and became direr by each day. On this alone, I have over 200 tabs open, scientific things another 370+… lol)
You should revisit the article and take the forecasting to its conclusion. We will live with lasting energy and supply shortages. Anyone in the medical field, working in a hospital, should talk to their supervisors and superiors (or anyone responsible for logistics) to secure a supply of any essential product that is made of plastic, and generic drugs. A following article is being written on this, but if you don’t want to wait, just revisit it.
Another day, another horror within our comprehension and in the parameters of forecasting. Before, a brief overview and explanation.
For the better part of 2025 and a portion of 2024, one of my research focuses has been the microvascular impact of SARS-CoV-2. Unlike the normal vascular system, the capillary system is a lot more delicate, significantly more intricated, harder to measure its health, harder to diagnose problems, but with an absurd impact on long-term health. When you use cutting-edge techniques and good data over the long term, you are able to predict outcomes.
Microvascular damage is often silent. It can be looked at as a literary physiological death by a thousand cuts. Microstructural damage accumulates over time, and it influences many diseases, rather than “normal” vascular damage, where dysfunction and pathology can be observed within a very short timeframe.
I have mentioned before in SARS-CoV-2 articles that sometimes you can do something akin to forecasting without all the complexity by simply performing a task of linguistic convergence. The same or similar words or terms showing up in different research papers within a shorter timeframe (a few months). And this occurred with microclots, microvascular damage.
At first, this impact was seen in a “small” percentage of Long Covid patients, but I knew from personal experience and observation of large amounts of data that, in reality, it was widespread. This was later confirmed (you can read about it in my neurology-centered articles, and dispersed on other articles) by analyzing the microstructural changes in the brain…than kidney…than liver…than elsewhere.
With extensive and cutting-edge research, we found that not all microclots are created equally. There are normal fibrinoid microclots, there are amylodogenic ones, and we found that extracellular proteins, released as a defense mechanism, also aid in the creation of these minuscule clots that are resistant to being dissolved by the body. You can get the most recent analysis below.
How neutrophils contribute to microclots
First and foremost. Thanks, everyone, for the birthday wishes, and for those who bought me a coffee as a gift, much appreciated.
For further context and as a reminder, Long Covid (and by sheer shared pathways, Long Vaccine) is a great tool to forecast wider populational dynamics of overlooked or under-researched physiological effects of the virus and its Spike Protein. And something that drove my research efforts for years.
Post-translational modifications within fibrinaloid microclot complexes distinguish Pre-COVID-19 Postural Orthostatic Tachycardia Syndrome (POTS), Long COVID, and Long COVID-POTS and reveal disease-specific molecular pathways
To be clear, the post-translational modifications measured here are related to our own proteins, such as fibrinogen, complement factors, apolipoproteins, etc, and not the Spike Protein. Spike Protein and its effects on our immune cells will certainly contribute to the formation of the complexes and the changes in the proteins, but they are not measured here. The pathology described, the PTMs, the amyloidogenic microclots, and the disease-specific signatures involve our own modified proteins.
There is one condition that many LC patients share with a few other patients, which is called POTS, Postural Orthostatic Tachycardia Syndrome, which is a fancy way of saying, when you stand, your heart beat spikes incredibly fast, it changes bloodflow at a systemic level and it causes dizzines, fatigue, fainting, and in some a hard time just living a normal live. Worth noting that the authors themselves mention that despite symptoms in LC patients, routine blood analyses will often show normal levels of common biomarkers and miss more delicated, hidden markers.
The authors named the microclots here FMC, which stands for Fibrinaloid Microclot Complexes. An important distinction is the complexes part. These are not simple clots but aggregates of multiple proteins. The group of patients in this study was separated into healthy controls with no SARS-CoV-2 infection or POTS history, Pre-Covid POTS (PC-POTS) with samples collected between 2015 and 2019, Long Covid without POTS (simply LC), and Long Covid with POTS (LC-POTS).
To visualize the shape (morphology) and quantify the microclots, they used the same approach, which involved using a dye that preferably attaches itself to amyloid structures. But to “see and measure” what is inside, normal proteomics (analysing many proteins in a sample), they needed to innovate, since normal methods would fail to measure, so they used a double digestion strategy to isolate the insoluble micro complexes fraction in the samples.
All disease groups, POTS, LC-POTS, and LC without POTS had FMC presence, confirmed by imaging, but there were no significant differences in the counts, meaning the quantity of microclots wasn’t enough to make a distinction between groups and their symptoms, and this was the reasoning for analyzing the proteins making the micro complexes. Only 3 proteins showed significant dysregulation in abundance across the groups.
MYH9 (involved in platelet function, cell motility, and structural integrity) was upregulated in Long Covid compared to controls
ZA2G (immune regulation, metabolism, and lipid degradation) was downregulated in LC compared to both POTS groups, but not significantly different from healthy controls
IC1 (inhibitor of core aspects of the complement/clotting cascade) was downregulated in PC-POTS compared to controls
If the quantity of FMCs and the number of proteins dysregulated aren’t enough to explain the symptoms and dysfunction, it strongly suggest that the pathological driver occurs at a post-translational level, meaning the changes occur after the protein is produced, which is why normal tests miss it, because normal tests only measure total protein concentration. Normal levels can be found, but the post-translational changes modify the protein into pathological.
At a PTM level, they observed extensive, disease-specific PTM dysregulation, choosing to measure peptides (very small proteins), where 29 unique base peptides from 16 proteins were regulated differentially, creating 64 uniquely modified peptide forms. 58.6% of these dysregulated base peptides were predicted by computer modelling to be highly amyloidogenic, cementing the connection between PTMs and the formation of pathogenic aggregates that are extremely hard for the body to dissolve.
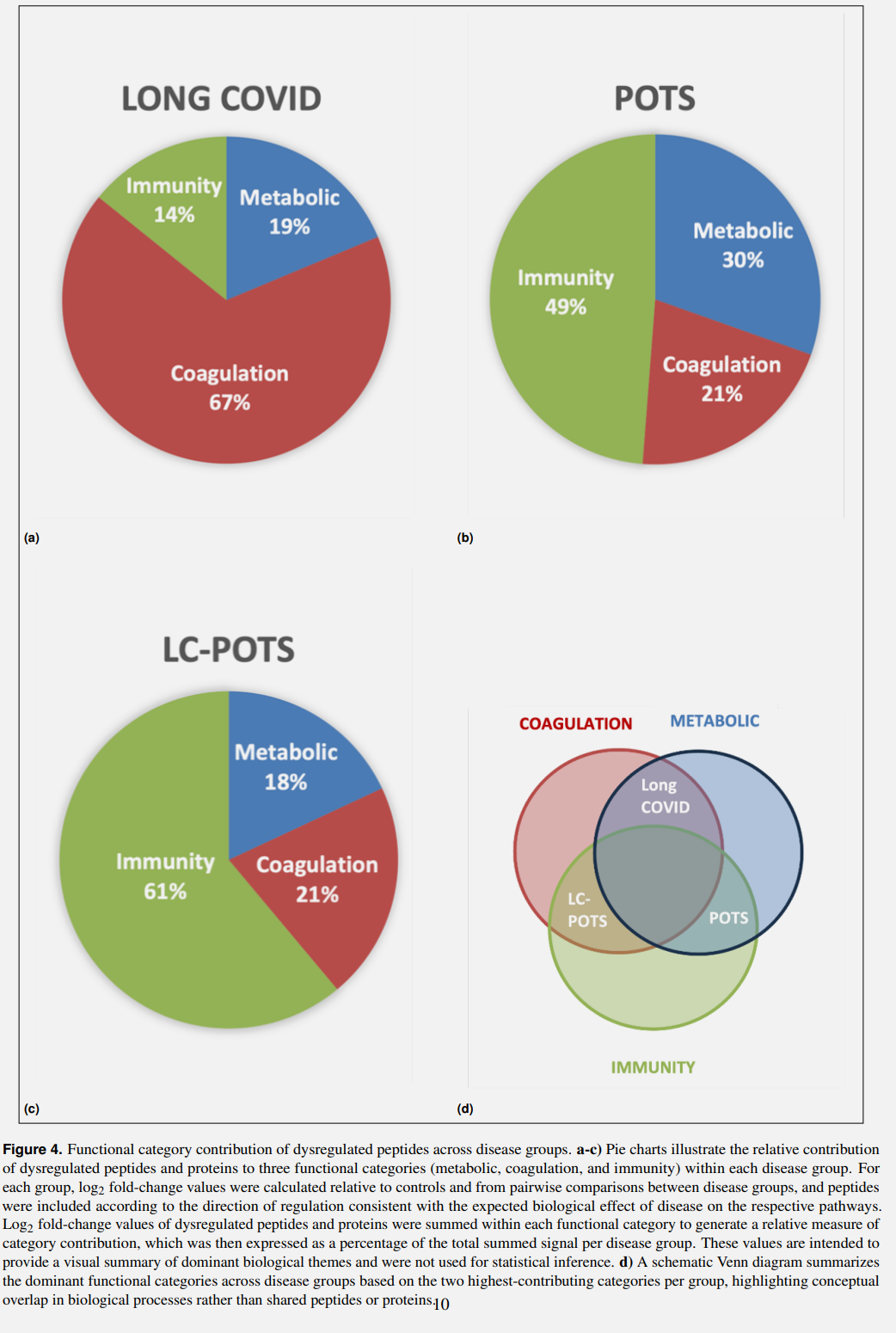
Each disease group had distinctive PTM changes in its FMCs and distinct pathways that contributed to the disease.
In Long Covid (no POTS), the main pathways dysregulated were coagulation (67% signal) with metabolic contribution (19%) . Fibrinogen peptides, especially the FIBA chain showed prominent AGE (Advanced Glycation End-products, the addition of small sugars to proteins and lipids, very harmful to cells) and the pattern here is highly reminiscent of the glycation seen in diabetes.
This is extremely significant for multiple reasons. First, SARS-CoV-2 is known to be a glycan “beast”, it uses these small sugar as a literal shield to avoid immune responses, not only to evade immune responses but to change how they occur. Even a mild infection impacts your glucose metabolism to a lasting degree, going from weeks to months, and of course, this form of metabolic dysfunction is present and observed in many of the Long Covid subgroups.
The AGE modifications here are induce by metabolic dysregulation rather than direct viral glycan transfer, but the infection-induced metabolic disruption likely creates the biochemical environment that favors these modifications.
This addition (modification) of sugar-derived adducts (AGE) to fibrinogen modifies it, forming bonds that are extremely hard to break, or to use precise framing, making clots and the PTMs extremely resistant towards fibrinolysis, which can lead to microvascular occlusion, leading to broader microvascular dysfunction over time because the FMCs trap inflammatory molecules and the body can’t break them down easily, inducing systemic, lasting inflammation. Of all feedback loops created by SARS-CoV-2, this is both a pillar to its systemic dysfunction and also extremely overlooked.
In Pre-Pandemic POTS (PC-POTS), the main pathways dysregulated are immune (49% signal), with a strong signal (30%) for metabolic dysfunction as a contributing factor. Distinct PTMs in complement proteins such as C3, C4A and B, and IGG1, with the downregulation of IC1. The protein A2MG was found to be upregulated in its oxidised and deaminated form (due to PTMs).
Dysregulated complement PTM suggests chronic, low-grade activation, downregulation of IC1 mirrors hereditary angioedema (a rare genetic disorder characterised by recurrent, severe, non-itchy swelling of skin extremities, such as face, hands, feet, but also the gut and airways). Complement activation is known to trigger mast cell activation (MCAS), which has a large clinical overlap with POTS.
Alterations in C3 and C4 are very important, both are central to both classical and alternative complement pathways. Oxidative modifications of some of their domains cause spontaneous hydrolysis and the creation of C3b-like activity, which depletes functional C3/C4 even though total protein levels appear normal. As a byproduct of these changes, C3a and C5a are formed (powerful anaphylatoxins), which drive Mast cell degranulation. They release histamine, causing quick vasodilation, tachycardia and orthostatic intolerance.
These changes lead to endothelial vulnerability due to metabolic alterations, chronic immune activation from complement and mast cells, together with the oxidative stress caused by the altered metabolism, which damages the endothelium, contributing to orhostatic intolerance that is a hallmark of classic POTS.
LC-POTS forms a bridge, or a sort of hybrid pathology. It is mostly mediated by immunity (61% signal), with secondary coagulation involvement (21% signal). It shares both the PC-POTS immune dysregulation in complement, IGG1, and A2MG PTMs, and also shares the coagulation pathology seen in LC without POTS (fibrinogen AGE modifications).
A2MG is as critical as the complement proteins because it acts as a molecular prison towards proteases (they break proteins down). Its bait region is cleaved by proteases, triggering a change in shape. Oxidation of a nearby tryptophan and deamidation of glutamine prematurely activate A2MG without protease engagement. This consumes A2MG, reducing its capacity to neutralize inflammatory proteases. A2MG normally binds IL-6 and TNF-α; modified A2MG loses this function, prolonging cytokine half-life.
This paper presents a unifying theory for distinct pathologies. Here, Long Covid is primarily a post-viral coagulopathy and metabolic-driven disorder. The viral infection induces severe oxidative and metabolic stress, leading to widespread protein glycation and oxidation, to which fibrinogen is susceptible. This creates amyloidogenic microclots that are resistant to being “dissolved” and disrupt the microvasculature systemically.
PC-POTS is mostly born from an immune and metabolic axis, a pathology with inherent dysregulation of the complement system, apolipoprotein metabolism, leading to chronic vascular inflammation, with added oxidative stress creating a feedback loop that damages the endothelium. This is why POTS has independent triggers, not necessarily needing viral infection (in theory, stress itself would be enough to induce it).
LC-POTS is a convergence of both. It is caused by viral-induced coagulopathy seen in LC, which is made significantly worse and lasting when there is pre-existing, or acutely triggered, immune-metabolic dysregulation. This combination leads to severe microvascular dysfunction manifesting as both post-viral sequelae and autonomic failure.
The hidden complexity
Now we do the usual, unifying a proposal or findings by intrepid researchers to my own observations, proposals, and writing.
AGE modification of proteins, or protein glycation, has significant, lasting consequences to the body, not only can they form stiff, resistant protein aggregations, but they also alter enzymatic activity, receptor signaling, and molecular recognition. AGEs are also DAMPs, damage-associated molecular patterns, danger signals to your cells. They can bind directly to RAGE, activate NF-kB, and trigger a pro-inflammatory, pro-oxidant, pro-fibrotic cascade.
This forms a glycoxidative state in LC and arguably a significant large number of people infected by the virus, albeit most recover after weeks. SARS-CoV-2 reprograms cell metabolism towards the famous Warburg effect, flooding the cell with triose phosphate intermediates, direct precursors for methylglyoxal, an extremely reactive compound, and a precursor for AGEs.
Infection and subsequent macrophage and neutrophil responses generate massive ROS via NOX enzymes. It wouldn’t be a problem, as the glyoxalase system (Glo1 and Glo2) detoxifies methylglyoxal. But Glo1 requires glutathione, and as I wrote multiple times, the viral infections basically drain glutathione from the body.
Do you care to remember what exactly binds directly with RAGE and interacts with AGE ? The one protein to rule them all, HMGB1. Direct binding to RAGE leads to sustained production of TNF-α, IL-1β, IL-6, NOX activation, thus massive ROS generation, and creates a self-perpetuating feedback loop of inflammation and oxidative stress, the exact environment that fuels PTMs.
HMGB1 can directly bind to fibrinogen, accelerating its conversion to fibrin, creating a denser clot. It induces Tissue Factor expression, thus activating the coagulation pathway. It inhibits fibrinolysis, and it is a potent NETosis inducer (FMCs are structurally associated with NETs). To add insult to injury, HMGB1 itself is heavily glycated and oxidized, both processes are PTMs of HMGB1 itself, the first enhances its pro-inflammatory activity substantially, the second is the dominant pro-inflammatory form.
By driving ROS via NOX, HMGB1/RAGE signaling accelerates the glycoxidation process, creating even more AGEs from circulating sugar and lipids. In turn AGE binding to RAGE on macrophages and endothelial cells triggers further HMGB1 release, creating a loop of HMGB1 → RAGE → ROS → AGEs → RAGE → HMGB1 release.
SARS-CoV-2 infection causes massive cell death (necrosis) and pyroptosis, releasing vast amounts of HMGB1. The initial viral spike protein may also activate RAGE. This sets off the HMGB1-RAGE-ROS-AGE cycle, with the glyoxalase system overwhelmed. The result is the dominant coagulation/glycation PTM signature observed.
The resulting storm of ROS, MG, and cytokines creates the perfect environment for the non-enzymatic PTMs (oxidation, deamidation, AGEs) that transform proteins like fibrinogen, complement factors, and apolipoproteins into amyloidogenic, aggregation-prone species. These aggregate into the persistent FMCs, trapping HMGB1, Spike, and cytokines, completing a self-sustaining circuit.
There are other pathways, and other proteins that contribute to the dynamics covered here, such as the Kynurenine Pathway, IDO1, Interferon-Gamma, but the one described above is substantiated and backed by a considerable amount of evidence (a couple dozen articles on my substack alone, and dozens of papers. Yes, plural).
I have long argued that fragments from the virus, be they genetic material (RNA), or proteins, especially the Spike Protein, would play a central role in long-term pathology, both from an acute (fast) to a slow, thousand cuts perspective. Protein complexes, as studied here, are incredibly impactful and important, as the evidence shows, they can play a disproportionate role in the symptoms and disease in an albeit stealth manner.
Both protein complexes formed by the virus and its systemic changes in the body, metabolism, cells and post-translation modification are incredibly important to understand and research as they will bear the answers to many of the “novel” effects not observed with other viruses, and they contribute to many of the effects I have covered in my Substack, such as immune cells dying throughout months after a mild infection. And I am mentioning fragments for a reason.
Thank you for your continued support and patience, especially during these dark times. It means a lot.





I wonder why all my spacing and formatting goes out of the window after I press "publish". I will never understand that.
Thank you for your patience, as I stated above, the reason behind the delay was simply focusing on the current affairs, as it has gone from bad to worse, and the fact I didn't have it in me to publish anything else (had a bit of the mind).
I have seen the impact in my own country with my eyes, and my government loves to sell the lie that we are "insulated" lol.
Interesting write up!
My daughter had POTS and it was debilitating. She wasn’t sleeping enough and then had a mild virus (we suspect covid- she had Delta and had a pretty good immunity so the case was mild) and then she got POTS. It was awful. We went to a pediatric cardiologist and she told us she would just need to live with it and it could take years! The doctor offered anti- depressants SMDH
We reversed it in about 6 months with the following protocol:
Slow walks slowly building up
Extra sun and vitamin d3
Extra iodized salt
Compression socks
Lots of salty pretzels
L-glutamine
Selenium
L-tryptophan
Methylated vitamin b complex with and emphasis on B2 and B12
She could hardly walk stairs it school it was so bad. Her worst periods she was nearly fainting and got these dark patches under her eyes- very scary.
I’m very thankful she’s doing better now and I hope maybe this info helps someone.
I think one doctor (whom I respect) theorized some POTs cases could be Vitamin B related because the nerves to the blood arteries don’t fire correctly and help pump blood back up to the heart and brain, so instead it pools by the feet (hence compression socks).