Microbiome, Long Covid, Insulin resistance and poor vaccine response
Tryptophan where you at, bro ?
Thankfully life came between me and writing this piece, multiple times, and that is a good thing for once because important papers were published within a few days of the first one, and a new paper within the subject bringing answers to old questions was also published almost every other day or so. We start on the paper I wanted to start with. Also yesterday was my birthday, so I decided to spend the time in the most useless way possible, as a sort of day off.
An underlying theme among many of my substack within this subject is the gut aspect of it all, especially the dysbiosis (moderate to severe shift in your gut bugs, a shift that will literally affect every single aspect of your body, for days to months and sometimes years. And of course a major “theme” in the past few months here.
Gut microbiota dysbiosis is associated with altered tryptophan metabolism and dysregulated inflammatory response in severe COVID-19
In line with previous evidence and even a rudimentary meta-analysis, authors found what is now a trend based on (almost) global evidence, that severe SARS-CoV-2 infection is often accompanied by a shift in the microbiota composition, often with the bad ones going through an overgrowth period, and the good/beneficial ones substantially decreasing in quantity, this, in turn, creates a shift in both immune responses, inflammatory processes, and many important metabolic pathways.
Here they found one of the good microbes that go through a significant drop was Clostridiales, and that oropharyngeal (your upper respiratory system, in a rudimentary way) changes are often due to antibiotic use. In the setting of this paper, the following was found:
COVID-19 severity was also associated with enhanced plasma concentrations of kynurenine, and reduced levels of various other tryptophan metabolites, lysophosphatidylcholines, and secondary bile acids. Decreased abundance of Clostridiales potentially mediated the observed reduction in 5-hydroxytryptophan levels. Moreover, altered plasma levels of various tryptophan metabolites and lower abundances of Clostridiales explained significant increases in the production of IL-6, IFNγ and/or TNFα. Collectively, our study identifies correlated microbiome and metabolome alterations as a potential contributor to inflammatory dysregulation in severe COVID-19.
I will first cover the rest of the paper, before interjecting with other research within the Clostridiales “theme”. As most readers will be aware (especially if you read the Kynurenine substacks linked above), the way your body metabolizes tryptophan can and will dictate much of your entire body, from how you generate “cell energy” to inflammation and your own immune system, and here they found, as backed by over a decade of evidence by now, a change on some microbes in your gut led to both a change in your tryptophan metabolites (metabolites are what is left after your body “digest” a molecule, grossly simplified), and thus a shift in inflammation.
Airway and intestinal microbiota disturbance in severe COVID-19
Taxonomic profiling further revealed that severe courses of COVID-19 robustly associated with lower abundances of several commensals including Ruminococcaceae, Lachnospiraceae, Bifidobacterium, Faecalibacterium, Roseburia, and Intestinibacter in the gut. Importantly, disease severity explained more significant variation in the intestinal abundances of these taxa than antibiotic intake during the sampling period. The depletion of commensals was exacerbated and in some cases potentially mediated by longer hospitalization times. Patients with longer hospitalization times additionally displayed depletions of e.g. Rotthia and Actinomyces along with increased colonization by Pseudomonas.
Each gut resident microbe has multiple functions, parts of these functions are to keep the bad microbes in check, and to effectively metabolize a sizable portion of your nutrients. An interesting point, and following something observed by critical care clinicians since 2020, was and is the abundance of Pseudomonas in severe patients, something that is “common” in many severe disease states, but way overrepresented in SARS-CoV-2 (it was the primary reason I started going down the bacteriophage angle in 2020).
The image, with prior substacks speaks for itself, in the line with previous research, there is a severe depletion and dysfunction of tryptophan metabolism in severe cases (and in Long Covid cases too, further down this), which creates a skewing to the Kynurenine Pathway, producing immune modulating metabolites, and shifting the inflammatory state of the patient, and here they point out that depletion of two gut microbes was correlated with these changes. I will quote the most pertinent parts of two extensive papers on the subject, I do recommend you to read them because all of this plays a much larger role than most give credit for in my opinion.
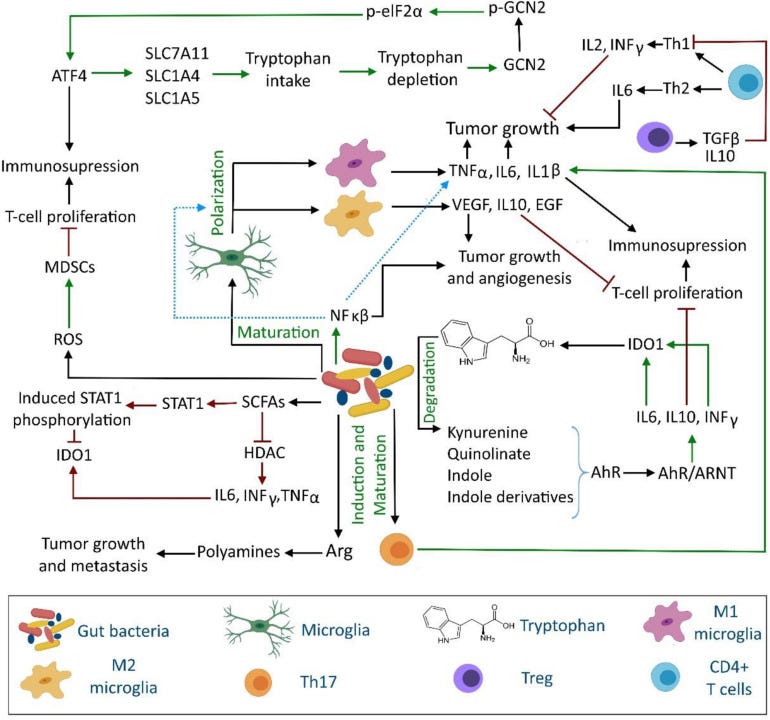
With respect of brain cancer, gut microbiota could modify the levels of antioxidants, amyloid protein and lipopolysaccharides, arginase 1, arginine, cytochrome C, granulocyte–macrophage colony-stimulating factor signaling (GM-CSF), IL-4, IL-6, IL-13, IL-17A, interferon gamma (IFN-γ), reactive oxygen species (ROS), reactive nitrogen species (e.g., nitric oxide and peroxynitrite), short-chain fatty acids (SCFAs), tryptophan, and tumor necrosis factor-β (TGF-β). Through these modifications, gut microbiota can modulate apoptosis, the aryl hydrocarbon receptor (AhR), autophagy, caspases activation, DNA integrity, microglia dysbiosis, mitochondria permeability, T-cell proliferation and functions, the signal transducer and activator of transcription (STAT) pathways, and tumor cell proliferation and metastasis. The outcome of such interventions could be either oncolytic or oncogenic.
I covered at least half of these here, and others covered them too and other aspects of it, and they all play an important role in the infection, and Spike-induced pathology.
It should be noted that the diversity, population, and metabolites of gut microbiota may control the state and level of both local and systemic immunity. The modulation of the immune system may be through the kynurenine pathway or through a direct effect on immune cells. For example, a bidirectional communication exists between gut microbiota and neuroendocrine system through the hypothalamic-pituitary-adrenal (HPA) axis
With respect to cancers, tryptophan depletion is increasingly being identified as a critical factor in tumor cell development. Importantly, tryptophan is highly catabolized in a microenvironment of tumor cells and inflammation regions.
It is worth mentioning that CD4+ T-cells show a reduced survivability in the presence of kynurenine under tryptophan-depleted locations. In tumor cells, abnormal angiogenesis is associated with the lack of blood supply, which is directly related to the development of hypoxia and the deprivation of glucose, amino acids, and other essential nutrients. Under this condition, the phosphorylation of eukaryotic initiation factor 2α (eIF2 α) occurs by activation of GCN2, which upregulates activating transcription factor 4 (ATF4) expression, increases amino acid biosynthesis, and eventually induces tumor immunoresistance and its survival.
Additionally, IDO-1 expressing tumor cells respond to tryptophan shortage by expressing amino acid transporter genes (i.e., SLC7A11, SLC1A4, and SLC1A5). The upregulation of ATF4-dependent expression of SLC1A5 and its splice variants eventually increase glutamine and tryptophan uptake that are highly demanded for rapid amino acids synthesis in tumor cells.
Expression of IDO-1 in non-antigen-presenting cells, such as tumor cells, promotes the escape of the tumors from immunosurveillance. Generally, IDO-1 can inhibit glucose uptake, glycolysis, and glutaminolysis, which contribute in its immunosuppressive activity
More importantly, gut-microbiota can affect the disease state of cancer by modulating IDO-1 activity (see Section “Kynurenine Pathway”) through regulating AhR activity. The interaction of some tryptophan-derived gut microbiota with AhR causes its activation. Ligand-activated AhR then regulates the functions of a wide range of innate and adaptive immune system such as dendritic cells, natural killer cells, macrophages, regulatory T-cells, and type 17 and 22 helper T-cells (Cheong and Sun, 2018). The dissociation of AhR from the chaperone heat shock protein 90 occurs after binding ligands to AhR. The ligand-activated AhR translocates nucleus and forms a heterodimeric complex with the AhR nuclear translocator (ARNT) protein. The AhR-ARNT complex is a transcription factor that regulates the expression of IL-6 in macrophages, IL-10 in natural killer cells and dendritic cells (Litzenburger et al., 2014; Wang et al., 2014). IL-6 then activates IDO-1 and indirectly contributes in increased kynurenine and kynurenic acid production and AhR activation. Moreover, AhR activation in natural killer cells induces INF-γ production that subsequently induces IDO-1 expression and eventually leads to tryptophan depletion (Figure 1).
Described in the paragraphs above are the highly complex biological pathway in which the body uses and regulates tryptophan and many of our physiological functions, even at a systemic level. It further explains and exemplifies some of the paradoxical dysfunction found in severe patients and persons with Long Covid.
The problem we find ourselves in is, this isn’t merely induced by the virus hijacking your cells for its own purpose, but the Spike Protein alone is able to activate and inflict most of these pathways, in this case creating negative feedback loops. The Tryptophan section ends up on a somewhat ominous note.
Overall, the depletion of tryptophan has pro-cancer activities (Table 1) and increases the survivability and severity of tumors. In a tryptophan-depleted microenvironment, the responses of antigen-specific T-cells are suppressed through accumulation of tryptophan-derived immunosuppressive metabolites (Mellor and Munn, 2008). As tryptophan is essentially provided through the diet, gut microbiota may help cancer cells to evade immunity in the human body through assimilation of available tryptophan in the gut.
Since I plan to cover other papers in this post alone, for brevity's sake, I advise you to read this paper titled Microorganisms, Tryptophan Metabolism, and Kynurenine Pathway: A Complex Interconnected Loop Influencing Human Health Status.
Increased insulin resistance due to Long COVID is associated with depressive symptoms and partly predicted by the inflammatory response during acute infection
Results. Long COVID is accompanied by increased HOMA2-IR, fasting blood glucose, and insulin levels. We found that 33.7% of the patients versus 0% of the controls had HOMA2-IR values >1.8, suggesting IR. PBT, but not SpO2, during acute infection significantly predicted IR, albeit with a small effect size. Increased IR was significantly associated with depressive symptoms as assessed with the BDI and HAMD above and beyond the effects of IO&NS pathways. There were no significant associations between increased IR and the activated IO&NS pathways during Long COVID.
Conclusion. Long COVID is associated with new-onset IR in a subset of patients. Increased IR may contribute to the onset of depressive symptoms due to Long COVID by enhancing overall neurotoxicity
This paper brings up interesting points and used a “fancy” way to measure the inflammation to explain these lasting metabolic changes in Long Covid patients, as I wrote on my Twitter, I wished they stopped measuring Long Covid solely based on severity, but also on viral load, but I get the reason why for the most part it must be severity based system.
All in all, there is only a small effect of inflammation during the acute phase on newonset IR, whilst activated IO&NS pathways during Long COVID appear to have no effect at all. This indicates that other factors related to the SARS-CoV-2 virus or COVID-19 are largely involved in new-onset IR. In this respect, evidence suggests that SARS-CoV-2 can infect and replicate in insulin-producing pancreatic β-cells, thereby resulting in impaired production and secretion of insulin and the metabolic dysregulation observed in patients with COVID-19 (60- 63) . Indeed, transcriptome analysis of infected pancreatic cell cultures confirmed that SARSCoV-2 hijacks the ribosomal machinery in these cells (64) . Microvascular damage caused by SARS-CoV-2 or via micro-thrombotic lesions may result in perfusion anomalies in pancreatic islets, which are required for glucose sensing and insulin release, whilst abnormal capillary architecture and fragmentation contribute to β-cell failure in T1DM and T2DM (65, 66) . As such, SARS-CoV-2 infection may cause direct pancreatic injury, which may worsen existing IR in T1DM/T2DM or contribute to new-onset IR and T2DM (67, 68) . Mechanistic explanations associated with COVID-19 are: a) COVID-19 is accompanied by a hypercoagulable state (69, 70) which may cause endothelial injuries leading to microvascular inflammation and thrombosis (71); b) autonomic dysfunction or autoimmunity (60, 61); and c) secondary mitochondrial dysfunctions (72) .
Besides all the myriad of factors that could affect insulin resistance in Long Covid cases, I would like to point out a few, that I previously mentioned, and some that I didn’t.
A few of what I could evidence I could keep sharing, this goes far beyond merely affecting the liver, Beta-Cells, or secondary damage, this is somewhat caused directly by how the Spike Protein interacts with your gut microbiome. For those who don’t remember, the Spike Protein can pierce most cellular membranes, it can burst biofilms (this will cause a flood of bacteria inside you), and effectively use the “skin” of bacteria (LPS) which is one of the most potent inflammatory molecules known to science to cause substantial changes and deeply affect you.
These 3 mechanisms alone can explain a lot of what we previously discussed, the changes and shifts in the microbiome, addressing both the microbiome/bacteria/gut health part, and the tryptophan dysregulation/insulin resistance are paramount for recovery of many of the LC, and people with persistent changes. Otherwise, you get a moment of relief and further aggression (another infection from Covid or other nasty pathogens) sends some of the more susceptible into the negative feedback loop. And this state of IR post-Covid is somewhat ironic. Why ? Because of this.
Reduced SARS-CoV-2 mRNA vaccine immunogenicity and protection in mice with diet-induced obesity and insulin resistance
Here is a few months old substack talking about the effects of poor glycemic control (insulin resistance) in relation to breakthrough infections, and delving a little deeper into the other aspects of how poor glycemic control affects your entire immune system, and immune response.
Inside you will find quite a prescient paper titled “How Hyperglycemia Drives Gut Barrier Dysfunction and Dysbiosis”.
In the paper above, the authors found that mice fed a diabetic diet (the respective to human diet is in fact high CARBOHYDRATE diet and not a high-fat diet) had a poor immune response when vaccinated with mRNA, in fact, the one particular type of cell that jumps to my eyes is the increase of CD8+, necessary for protection against severe disease and clearance of the virus was only seem in normal-fed mice.
Type 2 Diabetes, obesity, and other metabolic disorders all have a similar “cornerstone”, sharing similar mechanisms among themselves, and to the layperson sounding and looking exactly the same and they all are driven by the same physiological state, low-grade, persistent long-term inflammation, I have written about inflammaging, both alone and in regards to SARS-CoV-2 and the mRNA mechanisms.
The last line of the first highlighted paragraph is quite telling “short-term inhibition of inflammatory responses booster adaptive immunity in aged mice”, telling because the mechanism in which the Spike, mRNA-Spike in particular creates an immune response is highly inflammatory, beyond what one would expect a body to create to respond to a pathogen.
Finally a smaller piece of the puzzle, but one I wanted to know for a long-time. Not only do people with metabolic disorders have a weaker response to the mRNA vaccine (effectively making them merely spreaders of the virus) but the ones in most need are negatively affected by it, it is a multi-pronged physiological attack on your bioenergetic system at its very core.
Here the obese and diabetic mice had poor polarization to Th1, meaning they either end up in Th2, but most often (not measured here, as usual) Th17. Meaning this explain the worsening, or trending towards allergic states in many of the most at-risk population, but also the acceleration of certain pathologies and chronic diseases, without even giving you proper immunity against the virus. All risk, close to 0 benefits.
Since this is long enough, and with enough other substack mentioned that you should read, therefore making for complex, heavy reading, I will end this with another quick but positive thing I wanted to cover, I will leave the rest for the week since it would make this really long, one within the microbiome theme, the other about the paradoxical change in immune response in Long Covid, the microbiome one is so important it deserves a big chunk of text on its own.
A liver drug reduces SARS-CoV-2 entry into cells
A widely used drug called UDCA reduces SARS-CoV-2 infection in human organoid structures, animals and human organs maintained outside the body. Individuals using UDCA for liver conditions are less likely to develop severe COVID-19 than are people who did not use it. UDCA treatment could help to protect people with suppressed immune systems and offer protectionagainst vaccine-resistant variants.
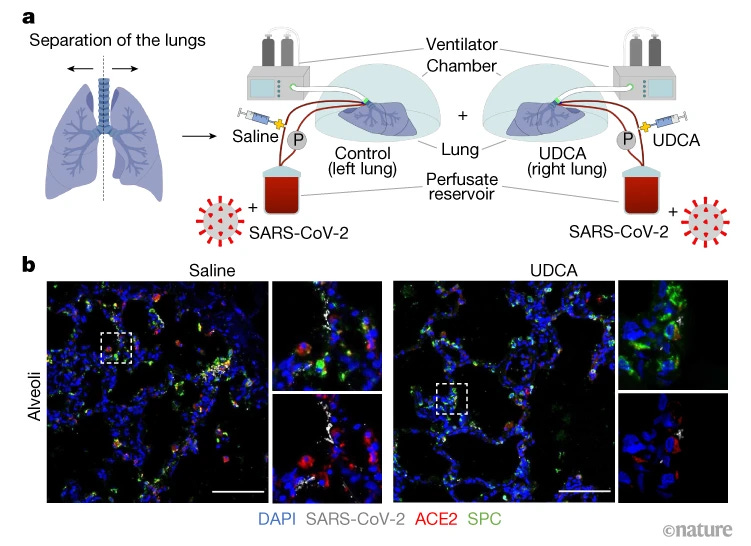
To confirm these findings in living animals, we treated hamsters with UDCA and showed that the drug prevented SARS-CoV-2 infection. To test whether these findings could be translated to humans, we tested UDCA in a pair of donated human lungs that were not suitable for transplantation. The lungs were ventilated and perfused with a blood-like fluid to keep them functioning, and then treated with either UDCA dissolved in a saline solution or just the saline solution, before infection with SARS-CoV-2 (Fig. 1a). UDCA treatment reduced SARS-CoV-2 infection of samples from the lungs (Fig. 1b). In eight healthy volunteers who received UDCA, ACE2 levels in nasal cells — the main entry points for the virus into the body — were reduced, in theory increasing people’s resistance to infection.
UDCA is a cheap, generic, off-patent, easy-to-produce (therefore scale) drug, with an incredible safety profile too. Here the authors found that the use of the drug can protect people from infection, and this type of intervention is still effective against the current vaccine-resistant variants and works in immune-suppressed people, a state untold millions find themselves in, without being actually aware of. The now common “being sick all the time”, or multiple infections within a short time frame, other states explain, but I am aiming here for simplicity, rather than complexity.
What really got my brain going here is that UCDA (Ursodeoxycholic acid) has many other interesting positive effects, counteracting aspects of the viral infection such as not only protecting the liber, but positively regulating amino acid, flavonoid, and fatty acid metabolism, in mice model it had the ability to regulate and restore the gut microbiome, repair the gut barrier integrity and attenuate hepatic (liver) inflammation, all aspects present in both vaccine-injured, the paradoxically immune suppressed, and the multiple times infected (another paper pointing to changes in microbiome upon the use of UDCA). It also has potent anti-inflammatory effects upon LPS exposure/stimulation.
Such changes in the gut, gut barrier, and immune responses and bringing back the balance in the microbiome, by itself, deserves more attention because as we just went through here, many of the severe/long-covid afflicted have the exact dysfunctions this cheap generic drug can potentially ameliorate or outright “fix”.
I will cite the following paper in the next microbiome SARS-CoV-2 substack, consider it a spoiler alert, a positive one. Incidentally, by bringing all these positive changes, but outside the parameters of most papers I went through, in theory via mechanistic analysis this drug can (most likely will) positively regulate and affect the kynurenine pathway, bringing it into proper balance.
Therapeutic role of ursodeoxycholic acid in colitis-associated cancer via gut microbiota modulation
Inflammatory bowel disease (IBD) is a predisposing factor for colitis-associated cancer (CAC). The association between bile acids and the gut microbiota has been demonstrated in colon neoplasia; however, the effect of ursodeoxycholic acid (UDCA) on gut microbiota alteration in development of colitis and CAC is unknown. Our analysis of publicly available datasets demonstrated the association of UDCA treatment and accumulation of Akkermansia. UDCA-mediated alleviation of DSS-induced colitis was microbially dependent. UDCA treatment significantly upregulated Akkermansia colonization in a mouse model. Colonization of Akkermansia was associated with enhancement of the mucus layer upon UDCA treatment as well as activation of bile acid receptors in macrophages. UDCA played a role in CAC prevention and treatment in the AOM-DSS and ApcMin/+-DSS models through downregulation of inflammation and accumulation of Akkermansia. This study suggests that UDCA intervention could reshape intestinal gut homeostasis, facilitating colonization of Akkermansia and preventing and treating colitis and CAC.
December 6 was my 1 year substack anniversary, and I truly appreciate everyone who chose or could keep supporting this work, also thankful for those who decide to buy me a coffee whenever they feel like it, and everyone who shares my Substack. Without all of you, this wouldn’t be possible.







Thanks John, another superb deep dive. UDCA noted.
Biome wise, if I can give a heads-up to quercetin:
Potential Implications of Citrulline and Quercetin on Gut Functioning of Monogastric Animals and Humans: A Comprehensive Review
...Quercetin possesses anti-inflammatory potential that can be expressed on different cell types, both in animal and human models [189,190]. Chen et al. [191] conducted an in vitro experiment to determine whether or not quercetin had the potential to inhibit inflammation in the small intestine of pigs by initially pretreating IPEC-J2 with quercetin, and then LPS. It was confirmed that pre-treatment of quercetin showed protective effects on the intestinal porcine enterocyte cells and inhibited porcine intestinal inflammation induced by LPS. Quercetin promotes mast cell stability, gastrointestinal cytoprotection, and also modulates gut immunity
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8621968/#:~:text=Effects%20of%20Quercetin%20on%20Gut,from%20colonic%20diseases%20%5B115%5D.
Multi-kingdom gut microbiota analyses define COVID-19 severity and post-acute COVID-19 syndrome
https://www.nature.com/articles/s41467-022-34535-8
Heavy reading indeed. I've skimmed for now but wanted to let you know I appreciate you pulling so many things together and posting for us. Our bodies are so amazingly complex I've often thought it nothing short of miraculous that we ever manage to survive at all.