Almost forgot, since I was raised by my mom. Happy Father’s Day to all the fathers who read my publication.
I guess my old observation will remain valid and meaningful for years to come…somehow. In a weird twist turn of events, a team of researchers has now provided evidence for yet another “novel” receptor for SARS-CoV-2. Keeping tabs on all of this is hard, so basically every few months a team of researchers discover that SARS-CoV-2 can bind to a novel receptor and explain a specific part of its pathology.
Highlighted summary of the article below.
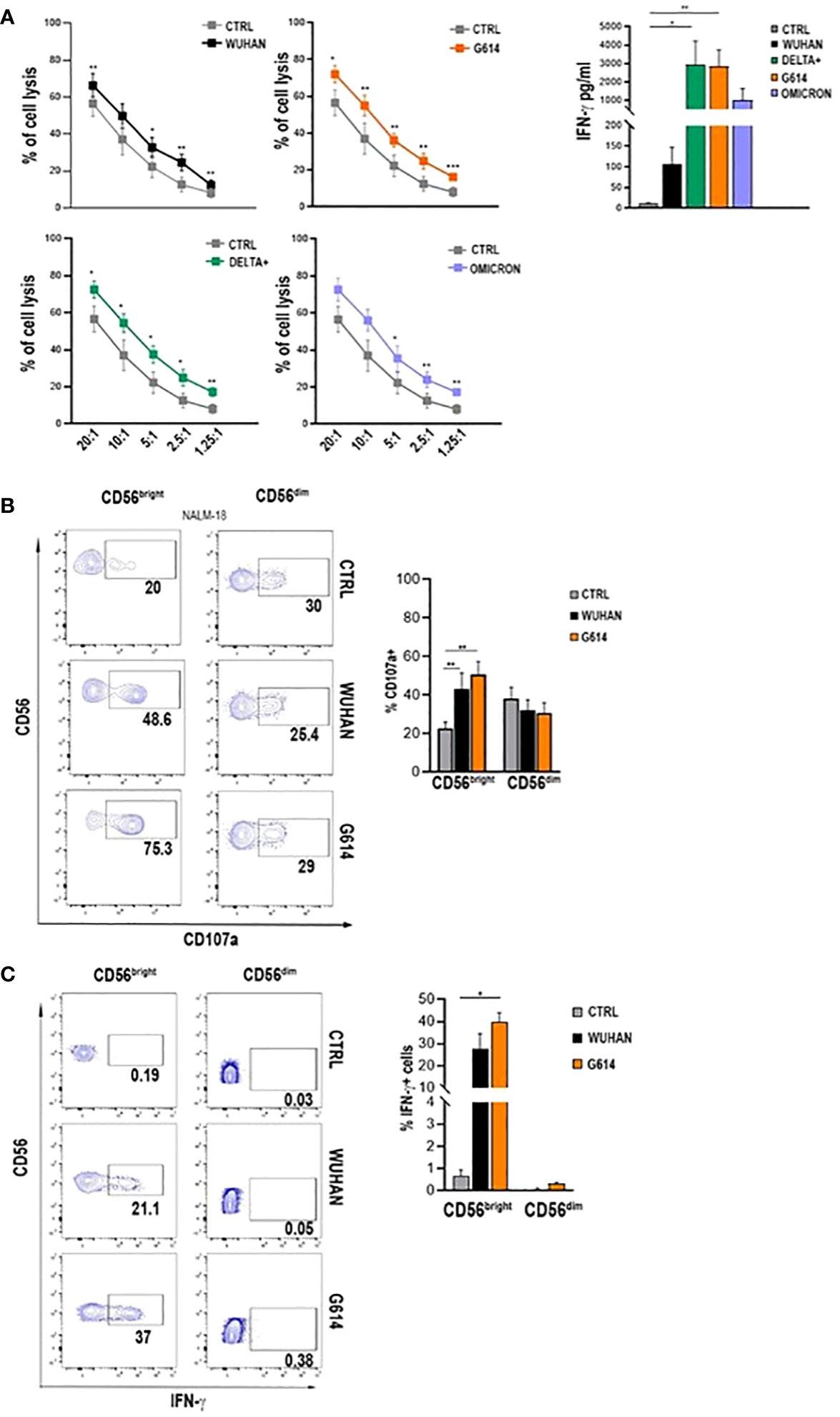
Spike Protein Activation: Recombinant Spike Protein (rSP) from various SARS-CoV-2 variants activates NK cells, marked by CD69 expression, with S1 being more effective than S2.
Functional Response: Activated NK cells, especially CD56bright, show increased cytotoxicity and Interferon-Gamma (IFN-γ) production; G614 and Delta-plus induce higher IFN-γ levels.
TLR Involvement: TLR2 and TLR4 are crucial for NK cell activation by Spike protein; blocking these TLRs inhibits NK cell activation and reduces IFN-γ production.
NK involvement in Spike Protein damage can be explained by this, among other complex immunological scenarios
TLR2/4 are novel activating receptors for SARS-CoV-2 spike protein on NK cells
The effects of SARS-CoV-2 infection on Natural Killer cells (NKs) are complex, there are significant differences between variants, and even between stages of the infection, but a widely recognized effect was depletion, often occurring as usual in severe cases with sometimes clinicians observing a loss of 50%+ of immune cells, but even mild cases suffer an atypical fluctuation in NK function and numbers. While many potential mechanisms were proposed, we lacked the evidence, until now.
Before diving into the study, a short explanation of Natural Killer cells is necessary, there are two main types of NK cells, CD56bright and CD56dim. NKbright is known for producing large amounts of cytokines thus it serves as a signaling cell, and orchestrates the immune response, while Dims are more specialized NK cells, they effectively are the ones that directly kill other infected cells and cancerous cells. Bright cells are like the military strategist calling and deploying forces, while Dim are the Special Forces executing direct attacks.
The first step to understanding what, how, or why NK cells are activated by SARS-CoV-2 is through looking at activation markers, these are often the many “CD-something” you can find in most immunological-centered scientific articles. You expose a cell to something, the cell has the “CD-something” which means it is activated, executing its function, and other markers mean it isn’t.
They exposed NK cells to recombinant Spike Protein (rSP) and while there was high variably, it was observed that rSP bound a similar proportion of both CD56bright and CD56dim (CD56 is used as a marker to differentiate Natural Killer cells). Next, they measured if the rSP could stimulate NK cell function, and no surprised, Wuhan Spike did in fact stimulate the function. A rather peculiar find was that they did not see apoptosis (cell death), so one of the remaining questions is, does only the viral infection kill cells ? Similar to how SARS-CoV-2 is able to infect lymphocytes but it is an abortive infection, the cell dies.
All variants tested here (Wuhan, G614, Delta-plus, and Omicron) were able to activate (CD69 presence) Natural Killer cells, but they were not able to affect the proportions of NK subsets, meaning Bright and Dim were similar (Bright is usually the most abundant one by a large margin) and recombinant S1, more than the S2 was significantly more effect at inducing NK cell activation.
This means the S1 part of the Spike is much more effective at interacting with NK cells than S2. These cells were more cytotoxic releasing more Interferon-Gamma, and among the subsets Bright NK cells displayed higher expression of CD107a and Interferon-Gamma than Dim cells. There was a variation between the amount of IFN-Gamma released from NK cells between variants, with G614 and Delta plus releasing more than Wuhan.
Their next step was uncovering which receptors are involved in the Spike Protein binding to Natural Killer cells and activating them, they first focused on ACE2, which they did not observe the expression during different times after stimulating the NK cells with Spike. Further, using neutralizing antibodies against ACE2 also didn’t affect activation markers. and neither the activation markers nor production of IFN-gamma was affected by doing the same with all major Natural Killer cell receptors (NKp46, NKp30, and NKp44, LFA1).
Well activation must be coming from somewhere, so they investigated the gene signatures, and found that genes related to the Toll-Like Receptor signaling cascade were highly upregulated in the simulated NK cells, and so they observed the phosphorylation of NF-kB. Both of these indicated that TLRs are being engaged.
rSPs from all VOCs bound both TLR2 and TLR4 with different binding capacity: Wuhan and Delta-plus rSPs bound TLR4 more than TLR2, while the inverse occurred for G614-rSP
By blocking TLR-2 and 4 using antibodies, they were able to inhibit Spike Protein from binding to fresh NK cells and also observed inhibited the expression of activation markers and education of IFN-gamma. Remarkably CD56 Dim NK cells from recovered patients respond more efficiently to Spike Proteins from different variants.
Among the author’s closing remarks, this one is the most significant one to me.
Of note, only one report indicated the direct activation of NK cells with two SP peptides through their binding to NKG2D (40). The presence of free S1 protein is a relatively frequent finding in a SARS-CoV-2 inflamed environment due to its release from virus-infected and apoptotic cells. This result highlights the importance of our findings and strongly suggest the involvement of soluble S1 in the pathophysiology of the disease. High plasma levels of S1 have been reported during severe disease, PASC, and post-vaccination side effects (17–19, 41), indicating that NK cells can potentially be stimulated in vivo throughout the entire course of infection. Lastly, the SP stimulation of NK cells in vivo can also explain the increased plasma and tissue levels of IFN-γ described as a very early event of infection (12) and initially attributed exclusively to virus-specific CD8+ T cells.
Among its novel findings, one of arguably great importance is the fact Spike Protein parts, with S1 binding with much more “strength” than S2 to receptors and activating NK cells. A very recent paper on acute myocarditis after mRNA vaccines found that NK plays a central role.
In a subset of Long Covid patients, there is observable dysfunction between lymphocyte function with high production of Interferon Gamma, and persistent pieces of Spike Protein interacting with NK cells would be another explanation. It would also explain why many experience a long-lasting systemic metabolic shift towards the Kynurenine Pathway.
IFN-Gamma is one of our body’s “napalm” recourses, and it is so potent, that it easily makes the cells produce IDO, the immuno-suppressing enzyme responsible for shifting cells towards the Kynurenine Pathway. This can create a dysfunctional loop that will affect response to cancers. (TNBC (breast cancer) cells induce dysfunction of NK cells through an IFN-γ/IDO-1/HLA-G pathway).
Adding to the complexity, LPS can, by itself, without engaging its main receptor TLR4, make NK cells produce IFN-gamma. LPS in and by itself can shift cell metabolism towards immune suppression after the initial onslaught of inflammation. A great review on IDO, Kynurenine pathway, and cell metabolism under inflammation-cancer states here.
I recently brought to attention the rather complex and reverse roles of both TIM-3 and Gal-9, which create an immune suppressed state when their expression levels increase. A common trend in a subset of severe and Long Covid cases is CD8+ dysfunction. “Natural Killer Cells Induce CD8+ T Cell Dysfunction via Galectin-9/TIM-3 in Chronic Hepatitis B Virus Infection”
Tim-3 is an inducible human natural killer cell receptor that enhances interferon gamma production in response to galectin-9 this will be important later since a new paper on Long Covid found high levels of Galectin-9 being produced without “apparent source”. Galectin-9 expression defines impaired NK cytotoxicity in patients with virus-associated solid tumors.
Other types of molecules will induce similar effects, on similar or different cells, often by the same mechanism. I will leave this old, but gold paper here.
“Lipid A, a derivative of endotoxin which does not contain polysaccharide, has similar antitumour action to dsRNA and endotoxin. Some common features of the chemical structure of lipid A and dsRNA are discussed.”
Besides my suggested supplements, it is one of the reasons Butyrate helps specific subsets of Long Covid, ME-CFS patients, “Butyrate limits human natural killer cell effector function”.
On a personal note and for my future self-reference, I hope soon we will have powerful enough tools (Machine Learning/AI models) to uncover which exact peptides (parts of the virus) are exactly enabling these endless “novel” effects.
And since we mentioned cancer…
One of my questions, left unanswered was always “What happens when a virus persistently and abundantly uses widely expressed receptors ? Does the body downregulate its expression, meaning lower the quantity present in cells ?” SARS-CoV-2 posses what I call “hierarchical receptor usage”, it basically binds to receptors and may change which one via complex interaction within the body (protein-protein interactions, immune system, etc).
ACE2 is the most well-known receptor, but as we just covered, it uses others, and its second favorite is TMPRSS2.
TMPRSS2 is a tumor suppressor and its downregulation promotes antitumor immunity and immunotherapy response in lung adenocarcinoma
Results
TMPRSS2 expression levels correlated negatively with the enrichment levels of both immune-stimulatory and immune-inhibitory signatures, while they correlated positively with the ratios of immune-stimulatory/immune-inhibitory signatures. It indicated that TMPRSS2 levels had a stronger negative correlation with immune-inhibitory than with immune-stimulatory signatures. TMPRSS2 downregulation correlated with increased proliferation, stemness, genomic instability, tumor progression, and worse survival in LUAD. We further validated that TMPRSS2 was downregulated with tumor progression in the LUAD cohort we collected from Jiangsu Cancer Hospital, China. In vitro and in vivo experiments verified the association of TMPRSS2 deficiency with increased tumor cell proliferation and invasion and antitumor immunity in LUAD. Moreover, in vivo experiments demonstrated that TMPRSS2-knockdown tumors were more sensitive to BMS-1, an inhibitor of PD-1/PD-L1.
Conclusions
TMPRSS2 is a tumor suppressor, while its downregulation is a positive biomarker of immunotherapy in LUAD. Our data provide a potential link between lung cancer and pneumonia caused by SARS-CoV-2 infection.
As I wrote shortly ago “It was estimated about 4,824,700 new cancer cases and 2,574,200 new cancer deaths occurred in China in 2022. Cancers of the lung, colon-rectum, thyroid, liver and stomach were the top five cancer types, accounting for 57.42% of new cancer cases.”
It will be interesting to observe how things change in the next 5 years… until the next one which will be about SARS-CoV-2 persistence.
If you choose or do support my work, I am grateful to you. You can also buy me a coffee =)



"Things hidden in complexity"... well titled. No one string to pull to unravel. Unfortunately.
... Which is why it would be a particularly stupid idea to use a 100% m1u modified agent that blocks both these PRRs in transfected DC's:
Collaboration of Toll-like and RIG-I-like receptors in human dendritic cells: tRIGgering antiviral innate immune responses
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3808934/