This is a rather shorter post, that will serve as a build-up to my Amyloid one, that will be lengthy and complex. You better start understanding these, and many other of my posts, otherwise you won’t understand why I call the amyloidogenesis, bullshit.
It will happen, just not how most of these people say it will, and not at a population level. Will cover some Long Covid papers, because they are important to also understand some of the dynamics going on, and vaccine injury.
First this small, but interesting paper.
Long COVID is associated with extensive in-vivo neuroinflammation on [18F]DPA-714 PET
First, to answer your question “what the hell is [18F]DPA-714 PET”. [18F]DPA-714 is a ligand, a biomolecule that binds to a receptor. In this case, this ligand has a high affinity to TSPO (Translocator Protein) mainly expressed in activated microglia. PET is the medical scan you probably heard about a couple of times in your life. In vivo, if you don’t remember, inside a living being, in the cases presented by this paper, two persons with Long Covid.
Summary
A significant number of COVID-19 patients develop ‘long COVID’, a condition defined by long-lasting debilitating, often neurological, symptoms. The pathophysiology of long COVID is unknown. Here we present in-vivo evidence of widespread neuroinflammation in long COVID, using a quantitative assessment, [18F]DPA-714 PET, in two long COVID patients. We reanalyzed historical data from three matched healthy control subjects, for comparison purposes. Both patients with long COVID had widespread increases in [18F]DPA-714 binding throughout the brain. Quantitative measures of binding (BPND values) were increased on average by 121% and 76%, respectively. This implicates profound neuroinflammation in the pathophysiology of long COVID
Results
Neuropsychological test and questionnaire scores
Neuropsychological test and questionnaire scores are provided in Table S1 and S2, respectively. Long COVID patient 1 had verbal memory deficits, mildly impaired sustained attention, presence of severe fatigue and concentration problems, and severe functional impairment. Long COVID patient 2 had visuo-constructive deficits, fluctuating sustained attention, presence of fatigue, severe concentration problems, and severe functional impairment.

Visualization is important, and I often forget about it (and sometimes just can’t find the proper way to show the readers something). From the data in the summary section, you have a visualization of what the 121% and 76% increase in the binding means. This paper is short but presents evidence to the building case that most of the damage in Long Covid is from neuroinflammation, which cascade into a systemic response as it often does. I would like you to take note of this paper about this ligand and Hepatic (liver) neuroinflammation, if needed make a digital or paper note of it.
Plasma Biomarkers of Neuropathogenesis in Hospitalized Patients With COVID-19 and Those With Postacute Sequelae of SARS-CoV-2 Infection
Results
We enrolled a total of 64 study participants, including 9 hospitalized patients with COVID-19 encephalopathy (CE), 9 posthospitalization neuro-PASC (PNP) patients, 38 nonhospitalized neuro-PASC (NNP) patients, and 8 HC subjects. Patients with CE were older, had higher pNfL and pGFAP concentrations, and more frequent pN Ag detection than all neuro-PASC groups. PNP and NNP patients exhibited similar PASC symptoms, decreased quality-of-life measures, and cognitive dysfunction, and 1 of the 38 (2.6%) NNP patients had pN Ag detectable 3 weeks postsymptoms onset. Patients with neuro-PASC presenting with anxiety/depression had higher neuroglial scores, which were correlated with increased anxiety on quality-of-life measures.
Discussion
pNfL, pGFAP, and pN Ag measurements indicate neuronal dysfunction and systemic involvement in hospitalized COVID-19 patients with encephalopathy. Detection of SARS-CoV-2 N Ag in blood 3 weeks after symptoms onset in a nonhospitalized patient suggests that prolonged antigenic stimulation, or possibly latent infection, may occur. Anxiety was associated with evidence of astroglial activation in patients with neuro-PASC. These data shed new light on SARS-Cov-2 neuropathogenesis and demonstrate the value of plasma biomarkers across the COVID-19 disease spectrum.
While I do believe in latent infection, and this will often be found in many of the Long Covid afflicted, I firmly believe there is prolonged antigenic stimulation present in many of them, for a multitude of reasons they can’t clear the viral fragments and persistent low-grade inflammation ensues, as I have mentioned many times recently.
Persistent clotting protein pathology in Long COVID/Post-Acute Sequelae of COVID-19 (PASC) is accompanied by increased levels of antiplasmin
Lingering symptoms persist for as much as 6 months (or longer) after acute infection, where COVID-19 survivors complain of recurring fatigue or muscle weakness, being out of breath, sleep difficulties, and anxiety or depression. Given that blood clots can block microcapillaries and thereby inhibit oxygen exchange, we here investigate if the lingering symptoms that individuals with Long COVID/PASC manifest might be due to the presence of persistent circulating plasma microclots that are resistant to fibrinolysis.
Fibrinolysis (anything with lysis, as a stupid rule of thumb) is the biological process of “diluting” or “digesting” fibrils. Lysis is the process, whatever word comes before it, is what is being “digested/dissolved”.
Patients who were more severely ill during their hospital stay tended to have more severe impaired pulmonary diffusion capacities and abnormal chest imaging manifestations, and are the main target population for interventions for long-term recovery.
In the current study, we investigate if we could find dysregulated molecules in circulation that might cause the lingering symptoms in individuals with Long COVID/PASC.
We have previously shown that large anomalous (amyloid) fibrin(ogen) deposits are present in plasma from acute COVID-19 patients [8, 9]. Here we show that plasma samples from Long COVID/PASC still contain large anomalous (amyloid) deposits, and that these deposits are most resistant to fibrinolysis, even with the treatment of a two-step trypsin method.
Of particular interest was a substantial increase in the acute phase inflammatory molecule Serum Amyloid A (SAA4) and α(2)-antiplasmin (α2AP) that were trapped in the fibrinolytic-resistant pellet deposit. The plasmin-antiplasmin system plays a key role in blood coagulation and fibrinolysis . Plasmin and α2AP are primarily responsible for a controlled and regulated dissolution of the fibrin polymers into soluble fragments.
Again, following a similar trend as before, where patients with more severe infections had more sequelae, this paper demonstrates the same. A portion of the people afflicted by Long Covid suffer from dysfunctional clotting, and their form amydoid-based microclots. The difference between normal clots and amyloid ones is simple. Amyloid clots are uncommon and very very hard to dissolve sometimes, especially if your body was overwhelmed by something else beforehand.
The researchers also found that these patients had poor plasma viscosity, meaning their blood was “thicker” than a normal person.
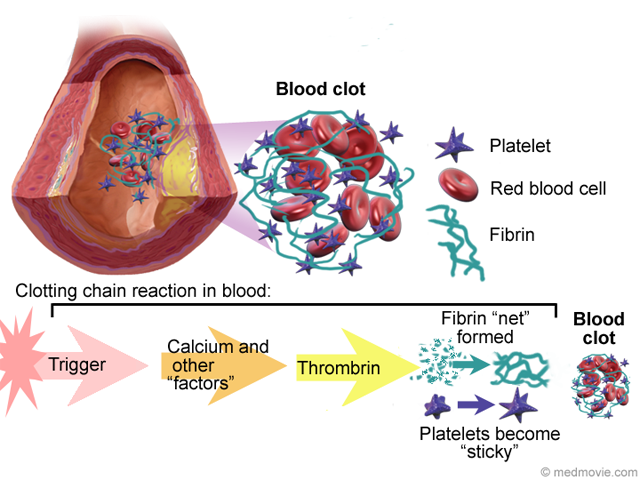
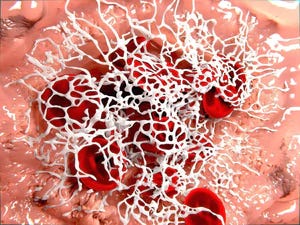
So why these clots are worse than normal clots. Better to illustrate it.
This is what a “normal” clot looks like, red blood cells, with a lot of platelets around and fibrin forming a huge “mesh” to plug any leakage, anywhere. An amyloid-based clot would look like this.
Your body won’t “digest” this clot as easily as a normal one, amyloid is a protein structure “made to last” in a very simplistic manner of describing it.
If you want a better understanding of what is going on in both of these papers and what might be doing it (news flash, it is the Spike, the S1 most likely) I wrote this quite a while ago.
The piece you are reading and both of the pieces I linked here are required reading if you want to have any understanding of the amyloidogenesis aspect of SARS-CoV-2 (vaccine, or virus, doesn’t matter), a post I decided to write, and will be (slowly) rework it and make a massive hypothesis out of it alone. Linking a lot of my work together.
As a clue to where this all leads.
SAA4 (also found as an apoliprotein of HDL), is synthesized constitutively in the >liver<.Here we report that SAA4 showed a significant increase in our proteomics analysis of the double trypsin-digested pellet deposits. There was a 17-fold increase in SAA4, between PPP in samples from Long COVID/PASC vs controls. We report fold changes in proteins present in the fibrinolysis-resistant pellet deposits of acute COVID-19 and Long COVID/PASC, compared to the fluid sample of the controls that also underwent a double trypsinization process. It follows that the true concentrations in plasma samples may therefore not reflect the fold changes we report on here, in proteins trapped in the solubilized pellet deposits. It was recently shown that patients with low HDL-cholesterol levels at admission to the hospital were more likely to develop severe disease, compared to patients with high HDL-cholesterol levels. Compared with the healthy controls, the patients have sharply decreased concentrations of total cholesterol, HDL-cholesterol and LDL-cholesterol. With reduced HDL levels in circulation, SAA4 potentially will be less partitioned into HDL.Of particular interest is the simultaneous presence of persistent anomalous (amyloid) microclots and a pathological fibrinolytic system.
I hope you all have a nice Tuedays !
If you are the anxious type, I already addressed how you deal with these problems here. Need to rework some a little and write a little bit longer explanations. Might do it the following days.



So, shorthand, they chose exactly the means in creating the "vaccine" that the virus uses to be so pathogenic?
“It was recently shown that patients with low HDL-cholesterol levels at admission to the hospital were more likely to develop severe disease, compared to patients with high HDL-cholesterol levels. Compared with the healthy controls, the patients have sharply decreased concentrations of total cholesterol, HDL-cholesterol and LDL-cholesterol. With reduced HDL levels in circulation, SAA4 potentially will be less partitioned into HDL.” I find this interesting. I am someone with high total cholesterol (at least in the eyes of the medical community), but definitely Pattern A (high HDL, low triglycerides, trig:HDL ratio of about 1). After after some troublesome side effects with Lipitor (CK skyrocketed) and Crestor (dizziness), I did some homework and now refuse to even discuss them. I wonder what role the overuse of statins based on faulty research that ignored all cause mortality (the model for vaccines?) plays here.