I decided not to send any e-mails, and in fact, just enjoy life on the weekend, so people could also live a life without worrying about the subjects we cover here. As a follower commented, “It felt like life was normal again, sports, beer, I was happy”.
This will not be similar to other pieces I published either by linking dozens of dots, or continuously linking a few dots and referring back to them. This is to inform the reader how the virus itself can be dangerous, and what all its proteins can do.
In fact, I recommend anyone with interest in SARS-CoV-2 as a whole to read the following paper in its entirety, I will just be using the “best parts”.
SARS-CoV-2: A Master of Immune Evasion
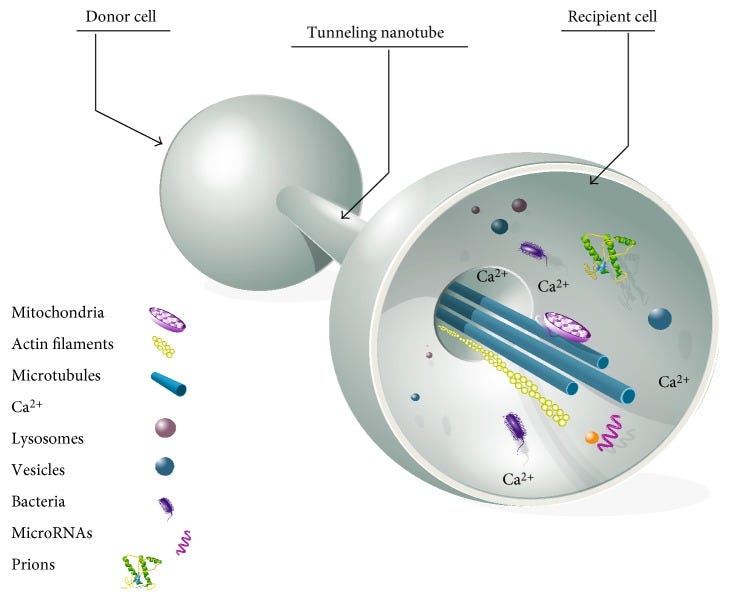
One of the virus’s immune evasion strategies is mitochondrial sabotage: by causing reactive oxygen species (ROS) production, mitochondrial physiology is impaired, and the interferon antiviral response is suppressed. Seminal studies have identified an intra-cytoplasmatic pathway for viral infection, which occurs through the construction of tunneling nanotubes (TNTs), hence enhancing infection and avoiding immune surveillance. Another method of evading immune monitoring is the disruption of the antigen presentation. In this scenario, SARS-CoV-2 infection reduces MHC-I molecule expression: SARS-CoV-2’s open reading frames (ORF 6 and ORF 8) produce viral proteins that specifically downregulate MHC-I molecules. All of these strategies are also exploited by other viruses to elude immune detection and should be studied in depth to improve the effectiveness of future antiviral treatments. Compared to the Wuhan strain or the Delta variant, Omicron has developed mutations that have impaired its ability to generate syncytia, thus reducing its pathogenicity. Conversely, other mutations have allowed it to escape antibody neutralization and preventing cellular immune recognition, making it the most contagious and evasive variant to date.
One aspect I was not aware of before the paper above was TNT, tunneling Nanotubes. Exosomes by themselves already make my head hurt sometimes. So here is a brief visualization of what they are/do. I think it is a mechanism important to bear in mind for the people interested in the subject because as you can see, it is incredibly diverse and important.
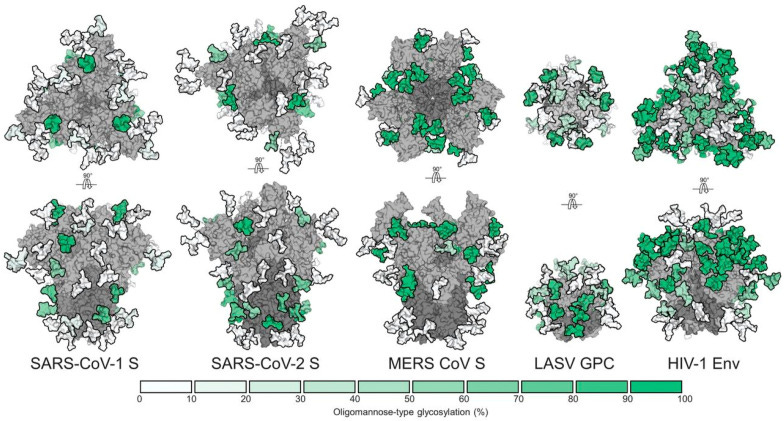
Spike Camouflage Employing Glycan Molecules (Epitope Masking)
Several viruses have adapted an epitope masking strategy by coating their envelope glycoproteins with glycans produced by the host, thereby preventing (or minimizing) antibody recognition.
Differential Impairment of MHC-I-Mediated Antigen Presentation by SARS-CoV-2 Variants
Viruses that cause long-term infections, such as HIV-1 and the Kaposi Sarcoma associated Herpes virus (KSHV), might elude immune surveillance by interfering with antigen presentation, which is necessary for immune activation, by decreasing the production of major histocompatibility complex I (MHC-I) molecules bound to the cellular membrane.
SARS-CoV-2’s open reading frame 8 (ORF8) produces a polypeptide that interacts with MHC-I molecules and causes their diminished expression. Infected cells with the Wuhan strain and with the Delta variant have a reduced vulnerability to cytotoxic T lymphocyte destruction because MHC-I molecules are specifically selected for their degradation within lysosomes (autophagy) in cells that express the ORF8 protein. Another study found that the ORF6 protein from SARS-CoV-2 disrupts the stimulation of MHC-I genetic expression
SARS-CoV-2-Driven Inhibition of Interferon Synthesis
A study found that RIG-I and MAVS interaction was blocked by the viral ORF9b protein from SARS-CoV-2, while the viral ORF7a protein disturbed the TBK1 protein, resulting in reduced IRF-3 phosphorylation, which is essential for type I IFN production
The SARS-CoV-2 ORF3a protein causes mitochondrial damage and mtROS liberation to enhance hypoxia-inducible factor 1 (HIF1-α) production, which enhances SARS-CoV-2 infection and cytokine release
However, if mitochondria are destroyed, the cell will not be able to perform other vital functions, such as the generation of energy through glycolysis or oxidative phosphorylation, so the term “mitophagy” is not correct. Therefore, the SARS-CoV-2 could impair only those selective mitochondrial pathways, which are related to the immune response, such as the inhibition of interferon synthesis.
The SARS-CoV-2 replication requires glycolysis (the Warburg effect, a modified form of cellular metabolism commonly found in cancer cells, where instead of the oxidative phosphorylation, aerobic glycolysis originating from the upregulation of several major glycolytic enzymes is used to efficiently produce ATP), and SARS-CoV-2-induced mitochondrial ROS generation activates HIF1-α, which then potentiates the expression of glycolytic genes and IL-1b
Again, to not make this a bible sized piece, I took the points that were of more interest to this one, and to the overall aspects, I covered so far. I have previously mentioned Glycosylation in a couple of other pieces, meaning per the description above, the use of very small pieces of “sugar” to glue and hide the places antibodies will attach themselves to, different viruses use this to different extents (clearly demonstrated in the image).
Glycosylation is entirely connected to glycemia, especially hyperglycemia, with too much sugar floating in your body. From the get-go, too much sugar floating around has only deleterious effects. Even one of the most inflammatory states your body can be (Th17 for a long period, or high levels in a short period), these T helper cells have a preferred metabolic state, and that state is a hyper glycolytic one. They just love sugar too.
And as per the paper above, each ORF has a modulatory effect on your immune system, with ORF8 affecting both your MHCs and also directly binding to the Interferon-Gamma receptor (basically affecting one of your most powerful antiviral responses). I will cover ORF6 in this piece soon.
If not clear, this is all in regards to the infection, and one of the mechanisms I kept referring to for months as “Omicron is Immuno suppressing everyone”, the more the virus replicates, the more these mechanisms come into play. From this paper.
ORF3a
All in all, ORF3a’s main interactions with the cell machinery have been described in inflammation, with IL1β secretion and NLRP3 activation, and in apoptosis.
ORF3b
Thus, SARS-CoV-2 ORF3b proteins showing significant activity against IFN-I are mainly localized in the cytosol, whereas their less active SARS-CoV homologues are found in both the cytosol and the nucleus.
ORF9c
Particularly, ORF9c expression impaired interferon signaling, antigen processing and presentation, complement signaling, and induced IL-6 signaling
SARS-CoV-2 type I Interferon modulation by nonstructural proteins 1 and 2
Different proteins can have redundant, even contradictory effects on your immune response, as shown by this very good paper. The closing statement is the main aspect to take from the paper for the layperson, which is simple to understand, SARS-CoV-2 modulates your immune system towards an inflammatory response, which should not be a surprise to anyone.
To understand how and why the modulation of your immune response will affect the outcome, this paper does a good job.
Interferon induction, evasion, and paradoxical roles during SARS-CoV-2 infection
However, the protective function of IFN is now well supported by the identification of human deficiencies in IFN responses as a predictor of disease severity. Here, we will review the cell types and pathways that lead to IFN production as well as the importance of IFN timing and location for disease outcome. We will further discuss the mechanisms that SARS-CoV-2 uses to evade IFN responses, and the current efforts to implement IFNs as therapeutics in the treatment of COVID-19. It is essential to understand the relationships between SARS-CoV-2 and IFN to better inform treatments that exploit IFN functions to alleviate COVID-19.
One such explanation is in the timing of these measurements. Humans are rarely studied immediately following infection and prior to the development of symptoms, and to the best of our knowledge, there is no study quantifying initial IFN levels in patients within the first hours/days of SARS-CoV-2 infection. This could represent a caveat because IFN production can be highly dynamic throughout the course of a viral infection. In animal models IFN levels dramatically vary at different times throughout a viral infection, reaching peak systemic concentrations within days after inoculation and reduced, or even undetectable, levels thereafter regardless of viral clearance or persistence.

And for last, and on purpose. ORF6.
Neurotoxic amyloidogenic peptides in the proteome of SARS-COV2: potential implications for neurological symptoms in COVID-19
If the proteome of SARS-COV-2 does contain amyloid-forming sequences, this raises the question, what is their function? It is known that viral genomes evolve rapidly and are highly constrained by size; therefore, every component is typically functional either to help the virus replicate or to impede the host immune system.
These ORF proteins were chosen as they have no obvious roles in viral replication26, perhaps freeing them up to have yet uncharacterised roles in disrupting the host antiviral responses.
We performed a bioinformatic screening of the ORF proteins to look for potential amyloidogenic peptide sequences. This analysis was used to select two sub-sequences, one each from ORF6 and ORF10, for synthesis. The synthesised peptides were both found to rapidly self-assemble into amyloid assemblies with a variety of polymorphic morphologies. Cytotoxicity assays on neuronal cell lines showed these peptide assemblies to be highly toxic at concentrations as low as 0.0005% (0.04 mg mL−1).
The cousin of this paper made the rounds and went viral months ago, to the point where the people pushing this line of thought got so much under my skin (for pushing stupidity and bullshit) that I started blocking away almost every single ago other people follow, refer to, or like.
Unlike the other papers, this one had the science done a little more properly, and with finding that is actually of concern to me. Per the author's juxtaposition, amyloids are not merely “bad folded protein bro”, they are a physiological response to “injury” at many levels, and so are prions for that matter. In fact, amyloids and other types of misfolded proteins (protein nanostructure) are an entire scientific field on their own, but that comes later.
Here the author used bioinformatics (mathematical models and lots of data) to look for potential amyloidogenic peptide sequences and found two. One in the ORF10, another in the ORF6. By using cutting-edge algorithms, they kept closing in on sequences that could fit the description, and in the end…
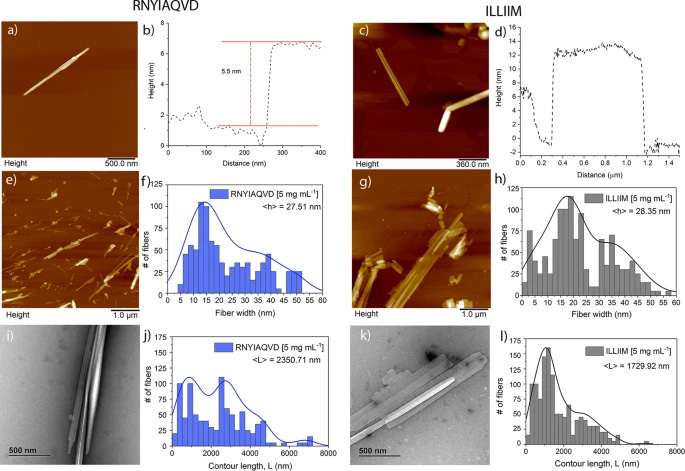
Based on the outputs from ZIPPER and TANGO and also on the experience in making and studying amyloid, we selected RNYIAQVD and ILLIIM to be synthesised and their amyloid-forming capability investigated.
For both peptides, the dominant polymorph was needle-like crystalline assemblies as seen in Fig. 2
Together this heterogeneous distribution of crystal heights provides further evidence for the polymorphic nature of both the ILLIIM and RNYIAQVD assemblies.
Such well-ordered 2D crystals were never observed for either peptide individually, and provide clear evidence of co-crystallisation. At this stage we have no evidence for the biological relevance of this co-crystallisation; however, as the ORF proteins from which these peptides are identified are themselves very small proteins (ORF6 is 61 amino acids in length), it is feasible that these small proteins may undergo similar co-crystallisation during their viral replication cycle facilitating an as yet unknown biological function.
To understand the paragraph above, the entire paper, and some of the more pertinent points of this paper you first need a quick note on fibrils and crystals. Protein (amyloid) fibrils are energy intensive in a way, they are more “disorganized” (3D shape is one of the most important aspects of protein folding and biology in general), while crystals are more organized and less energy intensive.
Arguably from purely a theoretical standpoint, the crystal would be more toxic, and harder to “dissolve”.
This is in particular supported by the previously reported neuroinvasive capabilities of SARS-CoV-27,8, the noted similarities of the symptoms to a (hopefully transient form of) AD5 and the previous detection of amyloid assemblies driven by other viruses20
This toxicity in relation to concentration is similar to that reported for Aβ4247 although expression levels and time-scales (sudden for COVID versus chronic for AD) are likely to be very different.
The significant increase in apoptosis and reduction in cell number seen for ILLIIM correlates with the work of Lee et al. who have previously shown that the ORF6 protein (that contains the ILLIIM sequence) is the most cytotoxic protein in the proteome of SARS-CoV-213.
Therefore, we hypothesise that the low energy ILLIIM crystalline assemblies are more slowly metabolised and cells are exposed for longer timeframes to the cytotoxic effect compared to RNYIAQVD assemblies.
The cytotoxicity and protease-resistant structure of these assemblies may result in their persistent presence in the CNS of patients post-infection that could partially explain the lasting neurological symptoms of COVID-19, especially those that are novel in relation to other post-viral syndromes such as that following the original SARS-CoV-1.
The ORF6 sequence (ILLIIM) crystal formation is more slowly metabolized, therefore cells in this experiment are under exposure for longer, leading to a more (biased I would say) toxicity profile, with a higher level of cell death and fewer cells available.
Before my last two-points, would like to bring a few points up, which the bs pusher often obfuscates.
This is a highly purified sequence, meaning the peptide sequence is isolated and introduced into cell cultures. For this to happen inside you, the body must cut down the ORF6 at the correct length most of the time, the virus has thousands of parts to be broken down.
While these papers have extremely valid findings, Cell culture and test tubes lack a few things. Like lysosomal function, ubiquitination, your immune system, influx and efflux of nutrients, and everything else.
And if you decide to dig into amyloid formation, you will find a common denominator at the core of the formation after the hit and run (the first injury that sets the formation off), for the amyloid to form in regards to SARS-CoV-2, you need dysfunctional redox system, especially one specific thing. I will let you guess.
Now to exemplify how complex amyloid and protein misfold truly is, especially in regards to infections.
When Host Proteins Coat Virus, Amyloid Fibrils Form
Viruses wrap themselves in a layer of host proteins.
The resulting “protein corona” makes a virus more or less infective.
When amyloid proteins are incorporated into the corona, fibrils radiate from the viral surface.
When a virus enters a biological fluid of a host—blood, lung, or cerebrospinal fluid—it becomes ensheathed by a unique set of proteins from its new environment. The proteins that make up the sheath differ depending on the fluid and on the type of virus. This “corona,” as Anna-Lena Spetz, Stockholm University, and Samir El Andaloussi at Stockholm’s Karolinska Institutet call the protein layer, shapes the interactions between virus and host cell. Besides rendering the virus more or less infectious, the corona incorporates amyloidogenic proteins. This changes their folding and spurs amyloid fibrils, which radiate from the viral surface, the authors report in the May 27 Nature Communications.
As my last point and a segway for part two.

Your metabolic state is truly what dictates your outcomes and potential to develop almost all diseases, especially neurodegenerative ones.
A very big thank you to all supporters here and those who use KoFi =) !








Forgot to add I will be sending another email shortly. Second one has be like LETS GOOOOOOOOOOOOOOOOO
Awesome work, and amyloid B involvement too in the KP. Helps contribute to the neurotoxicity.