Part one you should read before to make some sense of this, is below.
Very early on when Long Covid began to be taken seriously as a legitimate condition (I usually instantly block anyone who says otherwise, a sign of low cognitive capacity), hundreds, if not thousands of people were quick to point out the similarities between ME/CFS and Long Covid, with many experts and doctors pointing out it was basically the same disease.
I have argued they should rename both conditions to put them under the same umbrella. Recent papers now point out it is indeed the same disease, which I have agreed with since the start. The following is a rather extensive paper that I recommend anyone interested to read in its entirety.
Could the kynurenine pathway be the key missing piece of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome?
To investigate pathophysiology of ME/CFS, several potential pathologic hallmarks have been investigated; however, these studies have failed to report a consistent result. These failures in introducing the underlying reason for ME/CFS have stimulated considering other possible contributing mechanisms such as tryptophan (TRP) metabolism and in particular kynurenine pathway (KP). KP plays a central role in cellular energy production through the production of nicotinamide adenine dinucleotide (NADH). In addition, this pathway has been shown to mediate immune response and neuroinflammation through its metabolites.
A lot of the treatments for ME/CFS are symptoms based, in fact, most medicine itself is the treatment of symptoms and not the disease, creating what I call “pharma hostages” for life. Fixing people is not a profitable business…
One of the most prescribed treatments is antibiotics alongside other drugs, because at the core of the ME/CFS issue lies the chronic infection/long-term low-grade inflammation, and as we will see shortly, antibiotics are a short-term, poorly thought out stop-gap, that long-term will most likely worsen the symptoms. In the paper above, one of the promising treatments is rather… interesting.
Rintatolimod is a nucleic acid (double-stranded RNA) compound which acts as an inducer of interferon. Mechanism of action of rintatolimod is enhancing the NK-cell function and antiviral effects via influencing the 2-5A-synthetase [27]. Clinical trials reported that patients treated with rintatolimod showed improvement in fatigue (measured by exercise tolerance test) and quality of life in a subset of patients with CFS/ME, as well as postponing progression of the disease
While Long Covid/ME-CFS have a very complex root cause, in a big portion of patients, there is a common denominator if you do even a rudimentary form of meta-analysis. For different reasons (mechanisms) most of the afflicted suffer from dysfunctional immune responses. The second is even more interesting.
Another important immune modulator which has been studied in randomized clinical trials (RCTs) is rituximab. Rituximab as a monoclonal antibody is designed to reduce inflammation via bonding to CD20 receptors on B-cells and their depletion. It was initially administrated for non-Hodgkin’s lymphoma and autoimmune disorders
Anti-CD20 treatments are a promising form of mAbs treatment, per what is described in the paragraph above, and much more, and one thing CD20s does is modulate the expression of Th17 cells…Th17 are implicated in ME/CFS… Here is a very interesting paper on this subject.
As per the highlighted paragraphs, there are many correlations and similarities between what is going on with SARS-CoV-2 and other chronic infections. The 50 year old highlight is of special interest because isn’t what is described in that paragraph a simplified definition for inflammaging ?
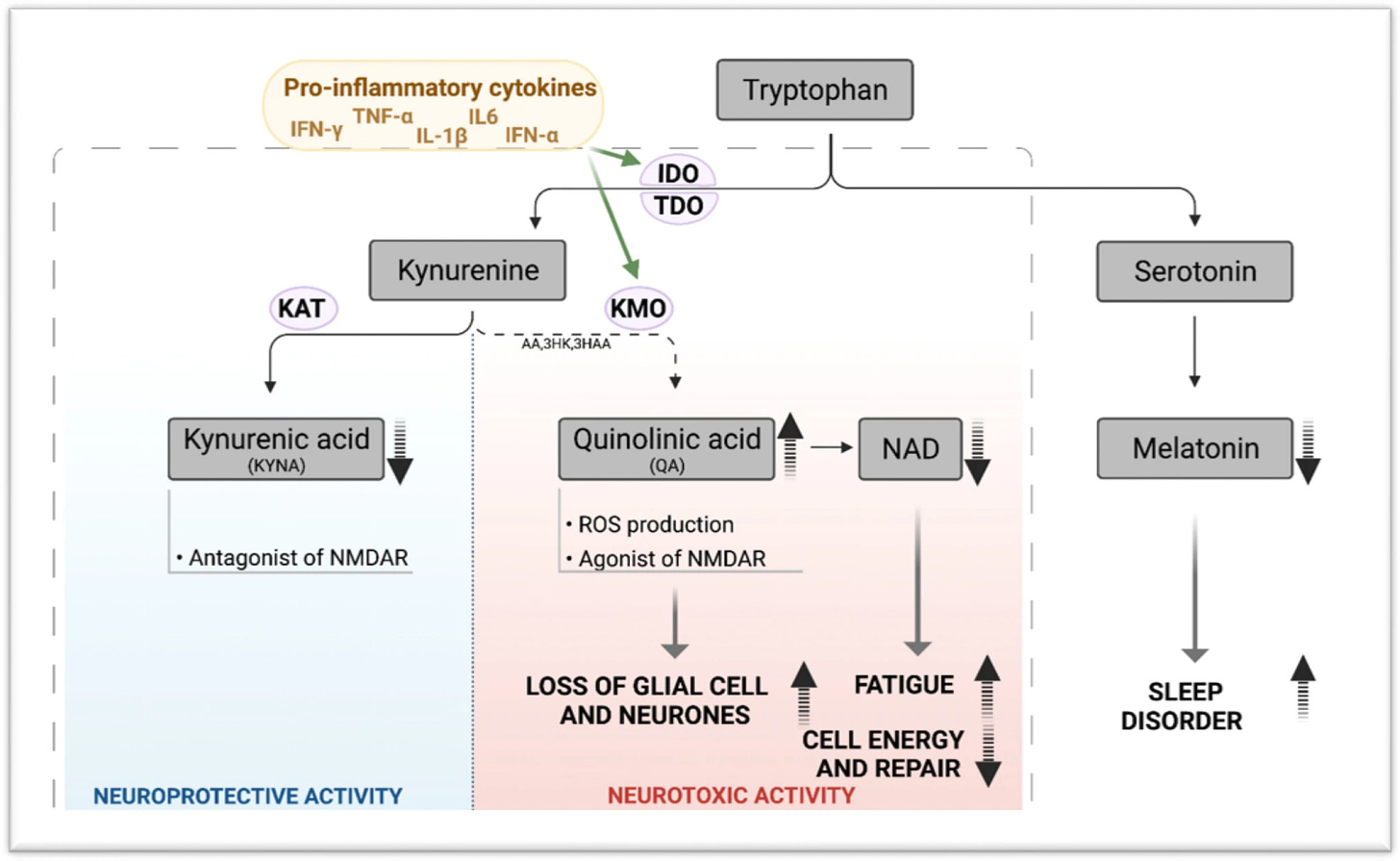
In a normal brain, TRP metabolism mediates crosstalk between blood, brain, and the immune system. Metabolism of TRP is tightly balanced between the activity of serotonin (5-HT) pathway and the KP. However, in the presence of pro-inflammatory cytokines, TRP metabolism is skewed towards KP and is activated to produce several neuroactive metabolites including the neuroprotective molecules such as KYNA and the neurotoxin QA instead of 5-HT and melatonin. QA has been proven to be involved in symptoms such as depression and suicidal behaviour in patients [129]. Given that depression has been reported in ME/CFS, QA can be studied as an effective excitotoxin contributing to neuroinflammation in these patients [130]. In addition to QA, NAD + as an important co-factor involved in cellular energy production shows some changes [131]. At first, by increasing QA levels under inflammatory condition, NAD + levels increase, however after saturation of converting enzyme and increase in oxidative activity induced by QA, there is a drop in NAD + production. Decreased levels of NAD + contribute to changes in several functions in cells, as NAD is involved in various cellular processes (e.g., calcium homeostasis, apoptosis, ageing, DNA repair, immunogenicity and transcriptional regulation) [132]. In addition, oxidative stress caused by increase in QA, can activate poly (ADP-ribose) polymerase (PARP), which repairs damage to DNA after oxidative stress. However, its overactivation leads to depletion of NAD + and ATP, consequently disturbance in energy production and mitochondrial function, which have been reported in ME/CFS as other pathologic conditions [133]. TRP metabolism and KP activity under inflammation have been demonstrated in Fig. 1.
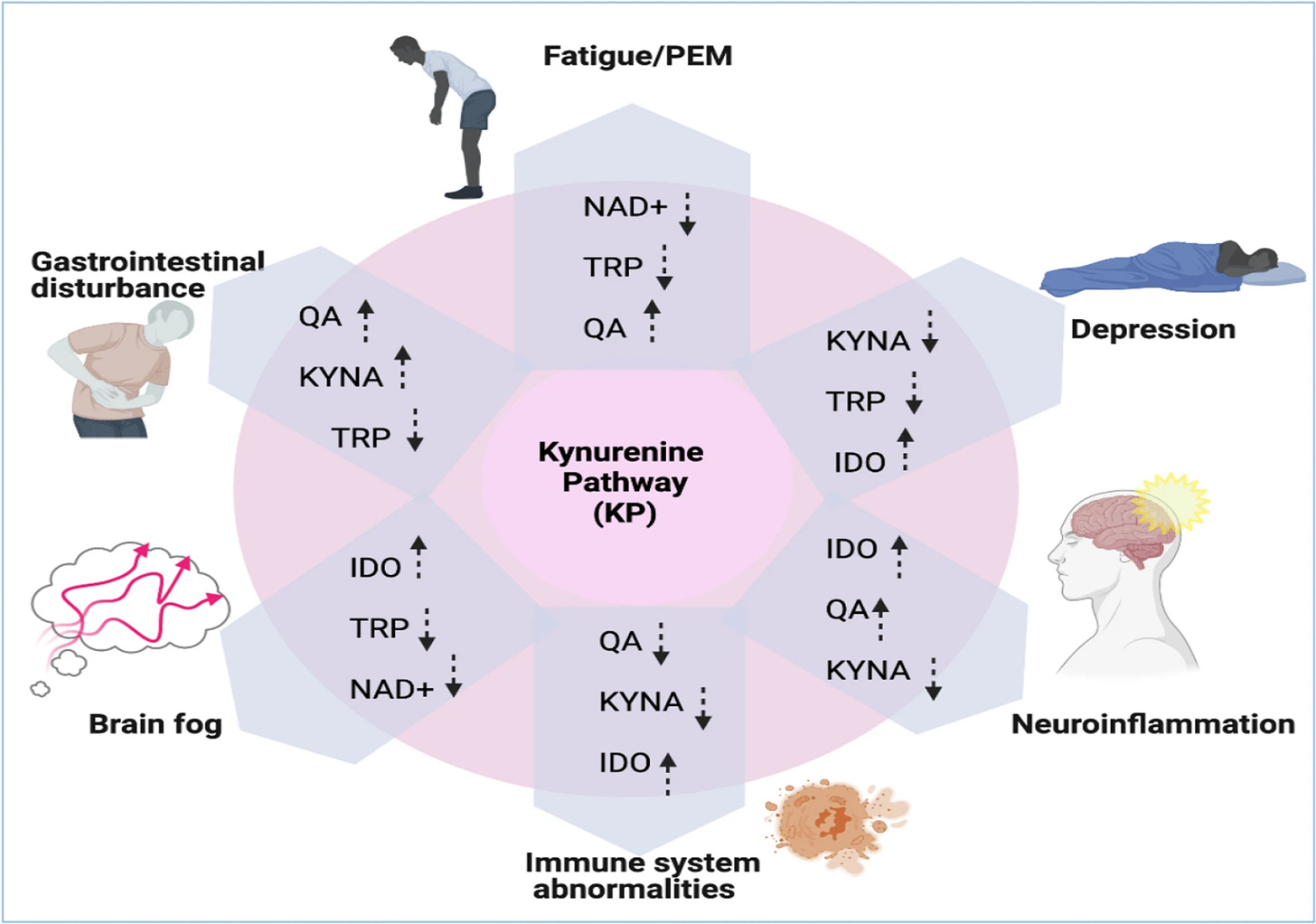
The Kynurenine Pathway is closely related to energy metabolism, therefore if there is a shift in the balance of its metabolites (byproducts of this metabolic pathway being activated) there are implications towards your whole bioenergetic state, in simple words, fatigue.
Either the dysfunctional state where your body doesn’t metabolize Tryptophan well or its depletion via many mechanisms (one of them being your literal cell activity and your immune response) will lead to everything in the image above. This is one of the reasons many people with states similar to, or with Long Covid will respond to anything that brings balance either to NAD+ metabolism or to Tryptophan.
But a skewed (disproportional) rate in Kynurenines and its metabolites will also lead to changes in your immune system and its balance.
SARS-CoV-2 infection can cause months-long changes in the glutamate metabolism, the changes it induces in your cells are enough to cause the state described here of immune suppression while still creating and incentivizing different levels of inflammation.
In line with everything we covered so far, there is a novel link between the KP and allergies, or allergic state and your gut microbiome.
Derangements in any of these TRP degradation steps can lead to altered metabolism of serotonin, melatonin, and KYN. Through the study of malfunctioning pathways, it is becoming increasingly apparent that TRP catabolism is a key player in the pathogenesis of gastrointestinal, cardiovascular, respiratory, allergic, neurodegenerative, and psychiatric disorders.
KYN Pathway Cross Talk Between the Gut Microbiome and Immune System
Residing in the human gut is our microbioata, a large and diverse collection of microorganisms which play a crucial role in regulating host and intestinal health (93). Bacterial cells within the whole human microbiome are more plentiful than human cells, especially in the gut, and their total gene count outnumbers that of the host by more than 100 times (94). The expression of this plethora of genes results in numerous enzymatic reactions with a myriad of physiological outcomes that would otherwise be unavailable to the host. It is for this reason that many now consider the microbiome to be an “organ within an organ” or the “second brain.” Not only is the gut microbiome essential for the digestion, absorption, and energy storage of food substrates, it also supports other immune and neurologic system functions (95).
In earlier studies employing a broad metabolomics approach, the composition of gut microbiota was noted to have a profound influence on circulating metabolites; plasma levels of KYN metabolites were affected to a greater extent than levels of other metabolites (96). In this study and ones that followed in germ-free rodents, plasma TRP levels were elevated in the pre-microbial colonization state, alongside reductions in serotonin and KYN levels (96–99). Serotonin, TRP, and KYN levels were normalized following microbial colonization of mice immediately post-weaning; normalization did not occur in rats. Similarly, Desbonnet et al.’s study of antibiotic-induced microbial depletion in mice reported higher circulating TRP levels and reduced peripheral KYN metabolism (92). Finally, changes to circulating KYN/TRP ratios have been observed following experimental induction of gastrointestinal inflammation via introduction of a parasite and likely altered gut microbiota (100).
The lowering of serotonin and KYN metabolite levels in the absence of gut microbiota and their restoration following the re-introduction of gut microbes indicates a key role for gut microbiota in the KYN pathway. More recent murine and human colonocyte studies provide further evidence for the regulation of gastrointestinal synthesis of serotonin by spore-forming microbiota, specifically by their metabolites, the short-chain fatty acids, deoxycholate, alpha-tocopheral, and others (101, 102). Of note, host TRP is required for this biosynthesis. How are these events initiated? We have already discussed the important role of AhR in modulating the immune system and the expression of IDO/TDO (103, 104). In the absence of AhR, endogenous KYNA levels have been documented to rise (105). TLRs are critical to the gut microbial community. In fact, activation of TLRs by microbial components has been identified as a key factor in initiating KYN metabolism. Several studies have linked microbial- induced KYN pathway changes to the expression of colonocyte TLRs in germ-free mice (99, 106, 107), namely, reduced TLR stimulation in germ-free mice has resulted in reduced TRP metabolism.
Your microbiome is literally the other half of your entire metabolic and immune health, any substantial change in the fine equilibrium in the microbiome will most certainly cause a shift in your metabolism, your capacity to produce neurotransmitters (up to 50% of the neurotransmitters are sometimes produced in your gut), some of the “stuff” you need are mostly or solely metabolized (made) in your gut by the good microorganisms there.
Therefore a change in your microbiome will inevitably lead to a change, among many others, a change in your Tryptophan metabolism, and a shift in how your body deals with whatever first initiated this response and with all the byproducts of the KP. While I didn’t write part 2 (for a few reasons, but the biggest one, I lost almost all the research in regards to it, need to start from zero) one thing is clear among the hundreds of papers I went through.
Kynurenine and its metabolites are situational and localized. A fair assessment is they are a double edge sword, in specific circumstances they can be protective, in others inflammatory or even toxic, a shift in the ratio (equilibrium between the quantities) influences the balance between protective>harmful. Even different regions of the brain will respond differently to its metabolites and difference in their levels.
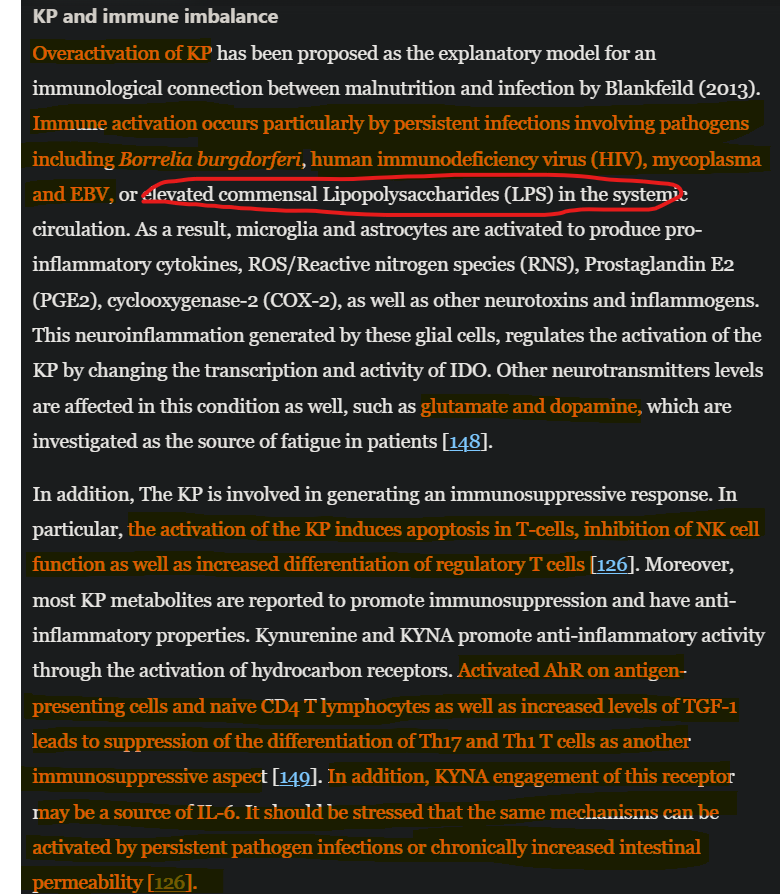
The quotes above exemplify exactly what I covered so far, when that fine balance is disrupted, you get all these deleterious effects. One thing in common among many, many chronically sick people, is often the undiagnosed, subclinical infection, bacterial or fungal.
Before I end with the paper that will link a lot of my entire Substack and most of the research I and someone else did, here are interesting points I want to raise.
Dietary Tryptophan Supplementation Improves Antioxidant Status and Alleviates Inflammation, Endoplasmic Reticulum Stress, Apoptosis, and Pyroptosis in the Intestine of Piglets after Lipopolysaccharide Challenge
Moreover, tryptophan downregulated the expression of ER stress (ER oxidoreductase-1α, ER oxidoreductase-1β, glucose-regulated protein-78, activating transcription factor 6, C/EBP homologous protein), apoptosis (B-cell lymphoma-2, BCL2-associated X protein, caspase 3), and pyroptosis signaling pathway (nucleotide-binding oligomerization domain-like receptor protein 3, caspase 1, gasdermin-D, apoptosis-associated speck-like protein containing a CARD). Collectively, tryptophan supplementation can contribute to gut health by improving antioxidant status and alleviating inflammation, ER stress, apoptosis, and pyroptosis in the intestine of piglets after lipopolysaccharide challenge.
There are similar findings in rats, therefore under a state of Tryptophan deficiency or poor metabolism (people will chronic infections and high levels of Interferon Gamma will be forever in a subclinical state of Tryptophan Deficiency, because Interferon Gamma regulates the use of Tryptophan, the KP, etc). From my own experience and helping people with many gut issues, some of them, with any type of dysbiosis or gut inflammation are usually poor Tryptophan metabolizers often responding well to the supplementation.
Melatonin ameliorates disease severity in a mouse model of multiple sclerosis by modulating the kynurenine pathway
Something that I didn’t mention much, but a lot of people afflicted with Long Covid and ME/CFS would benefit greatly from Ozone therapy, which is fairly cheap and available anywhere in the world. Ozone therapy allied either with high dosages of certain supplements or IV treatment of Glutathione, Vitamin C, NAD+ and Complex B (best option, pricey though) often sees a great improvement, and supplementation takes care of the rest. Paper on Ozone therapy here.
Results: Almost half (43.5%) of the treated patients evolved their FSS scale from the worst (7) to the best (1) score, assessing the highest improvement from being treated with O2-O3-AHT. Furthermore 77.5% of patients experienced significant ameliorations of fatigue, of 4–6 delta score. No patient showed side effects, yet experienced long lasting fatigue disappearance, by three months follow up (4) Conclusions: Treatment with O2-O3-AHT greatly improves ME/CFS related fatigue, aside from sex and age distribution.
With these out of the way…
A paper was recently published about how Spike Protein binds to LPS. No, not the one that I covered a couple of weeks ago, a new one, with much more evidence. It answers A LOT of questions.
LPS induces hypoxia-inducible factor 1 activation in macrophage-differentiated cells in a reactive oxygen species-dependent manner
To understand more about HIF-1a. Among many others.
Appreciate all supporters who enable me to do this, and subscribers who share ! Thank you !








Always interesting to see how the body systems are so interrelated. Especially the digestive system impacting so much else.
Thank you, love the biochemistry John paul. Cfs/me is a topic of podcaster betterhealthguy. he interviews practitioners about their treatments for many chronic conditions.