I stayed away from this paper for a few days, had internet problems during the weekend, and I was waiting if someone else would cover it.
Accelerated biological aging in COVID-19 patients
We also find the increasing acceleration of epigenetic aging and telomere attrition in the sequential blood samples from healthy individuals and infected patients developing non-severe and severe COVID-19. In addition, the longitudinal DNA methylation profiling analysis find that the accumulation of epigenetic aging from COVID-19 syndrome could be partly reversed at late clinic phases in some patients. In conclusion, accelerated epigenetic aging is associated with the risk of SARS-CoV-2 infection and developing severe COVID-19. In addition, the accumulation of epigenetic aging from COVID-19 may contribute to the post-COVID-19 syndrome among survivors.
Abstract pretty straight forwards, they epigenetic aging (genetic aging without affecting your genes), and telomere attrition (shortening) in healthy people, and infected patients at different degrees of severity. They propose that the accumulation of this “molecular biological aging” contributes to Covid after effects.
Aging is a biological process related to diseases and mortality. The biological process in aging is reflected by molecular hallmarks, which include epigenetic modifications and telomere attrition9,10,11. DNA methylation correlates with aging process and can be used to estimate epigenetic aging across tissues12,13,14. The deviation between DNA methylation age (DNAm age) and chronological age has been proposed as a biomarker for aging and has been related to risk and survival outcomes in age-related diseases15,16. Previous work showed that the epigenetic landscape of host cell is altered during HIV17,18,19 and coronavirus20,21,22,23 infection, including SARS-CoV-2. Interestingly, telomere length and the epigenetic clock do not correlate with one another, suggesting that DNAm age and TL measure different aspects of biological aging28,29.
There are extensive videos, tutorials, and lectures about DNA methylation on the internet, and it is for me the most boring aspect of biology, and medicine above ALL, I think only epidemiology is more boring (and outright useless). Also find it intriguing that finally, some researchers propose telomere length, and epigenetic clock (your biological age, not your chronological, how old you are, you can be 50 years old, but biologically 40). They go on to explain how they measured it.
DNAm - DNA Methylation TL - Telomere length
Accelerated epigenetic aging in SARS-CoV-2 infection
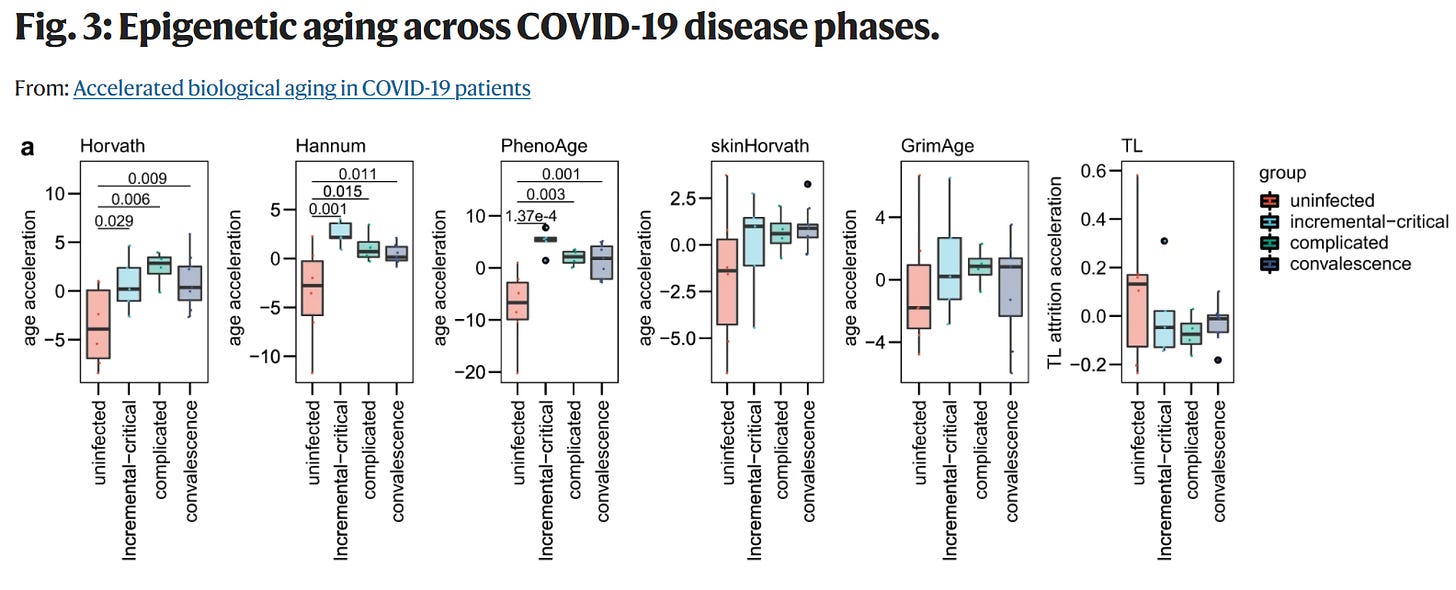
We initially assessed the DNAm ages and TLs of the blood samples and found an older DNAm age in the COVID-19 patients for Horvath, Hannum, skinHorvath and GrimAge clocks compared to the healthy individuals. To adjust for the bias due to individual chronological age, we calculated epigenetic age acceleration for each sample. Individuals with COVID-19 were estimated to have significant DNAm age acceleration for Hannum, PhenoAge, skinHorvath and GrimAge clocks and significant DNAm TL attrition acceleration compared with healthy individuals
Using 5 different methods to measure the DNAm, and TL, they found that COVID-19 patients had older DNAm age. Pretty straightforward so far.
Accelerated epigenetic aging and risk of developing severe COVID-19
Next, we analyzed the association of COVID-19 severity with epigenetic aging. We found individuals with severe COVID-19 had significant DNAm age acceleration for all epigenetic clocks and DNAm TL attrition acceleration compared with healthy individuals. In addition, severe COVID-19 patients had significant DNAm age acceleration for Horvath, Hannum, PhenoAge and GrimAge clocks and significant DNAm TL attrition acceleration compared with non-severe COVID-19 patients . Moreover, non-severe COVID-19 patients had significant DNAm age acceleration for Horvath, Hannum, skinHorvath and GrimAge clocks and significant DNAm TL attrition acceleration compared with healthy individuals . Among the five epigenetic clocks, the statistical difference in the pairwise test for GrimAge clock was most significant. In addition, we found that COVID-19 patients developing pneumonia had significantly accelerated epigenetic aging compared with those not developing pneumonia . Together, we found an increasing acceleration of epigenetic aging in the sequential samples of healthy, non-severe and severe groups.
As you would expect, the severe disease leads to a significant acceleration of this specific type of biological aging. And comparing severe to non-severe patients, severe had more than non-severe, which is to be expected, your body is overwhelmed, stripping resources from everywhere it can to fight, stay alive, and correct all the dysfunctions going on. Non-severe individuals, compared to healthy ones had more aging too. So basically, once infected, your body ages because the virus is hijacking your cellular machinery, wreaking havoc, and creating “cellular trash” your body is overwhelmed by it.
Here is a really good paper on this. Oxidative Stress in Cognitive and Epigenetic Aging: A Retrospective Glance, and the following from this paper.
Mitochondrial dysfunction plays a role in determining epigenetics changes during aging. Increased ROS leads to changes in the methylome, inducing aspects of the epigenetic aging process (Kietzmann et al., 2017). ROS causes DNA lesions and the most common base lesion is 8-hydroxyguanine (8-OHdG), which is in fact used as a measure for the level of oxidative stress (Turk et al., 1995). 8-OHdG lesions in cells caused by ROS inhibit DNA methylation, in line with the hypothesis that mitochondrial dysfunction and ROS production may be a driver of altered DNA methylation observed during aging
The reason I am sharing this in the middle of the post, not at the end, is because most people tend to “lose it” with certain topics, this being one of them. It is entirely reversible, you get “most of the years lost” back in a way. Of course, it is multifaceted and complex, and numerous other variables come into play. A couple of posts covering the Mitochondrial Dysfunction, and another here.
Similarly, Hannum and PhenoAge clocks were accelerated at the initial stage of incremental and critical disease phase and reversed in the upcoming complicated and convalescence phases. In addition, an increasing attrition acceleration of DNAm age at the initial two disease phases was found to be partly reversed in the upcoming convalescence phase, although the differences between every two phases were not statistically significant.
Furthermore, we tracked the dynamic changes of DNAm ages and TLs during the clinical disease phases in individual patients and observed similar result to the group-wise comparison. The accelerated DNAm ages at initial phases were partly reversed at the later phases in some COVID-19 patients by using the five epigenetic clocks, while the reversible outcome of accelerated attrition of DNAm TL from initial to later phases was found only in one patient. Therefore, we speculated that COVID-19 syndrome might accelerate epigenetic aging in SARS-CoV-2 infected patients, and this process would be partly reversible in the clinical disease phases.
To reiterate, they found accelerated DNAm age, and it reverses by itself at stages of the infection, and everything I discussed so far. This isn’t exactly new, as the paper itself references a similar mechanism in other viral infections.
I won’t go into details right now on how to avoid this because it will be in my amyloid post, just see the post below, it also addresses it, especially the one about how to minimize Spike damage. If you want to dig yourself, senolytic compounds.
You can buy me a coffee whenever you feel like it.
Deep appreciation for all the supporters!



Has this kind of analysis ever been performed for other viral diseases e.g. influenza? It would be helpful to know how Covid compares to other viral diseases to which we are typically exposed...
Thanks, an important topic to keep investigating. Bioweapon is what bioweapon does.